Research on the Adsorption Performance of Microwave Combined with Alkali Modified Fly Ash for Cr(Ⅵ)
-
摘要:
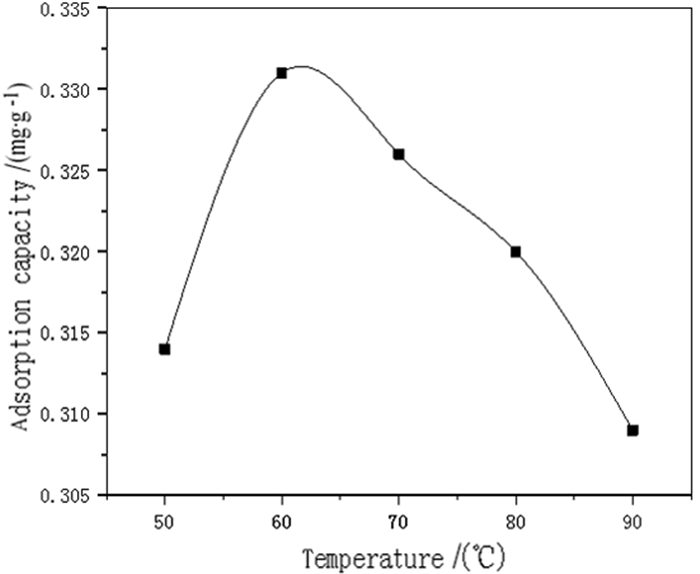
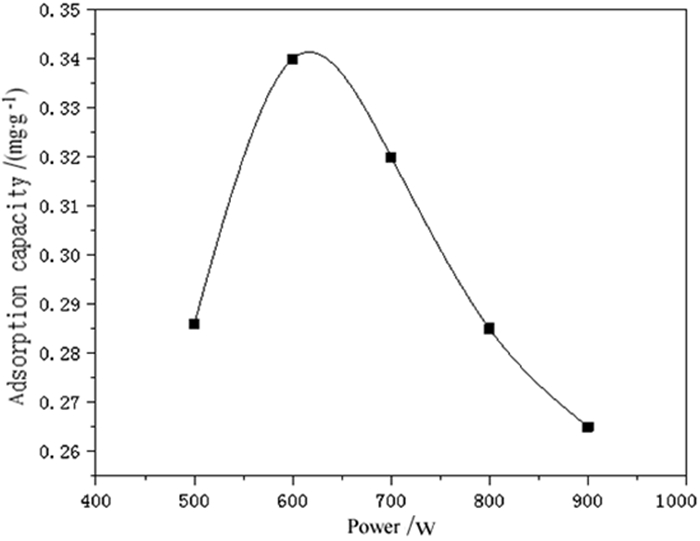
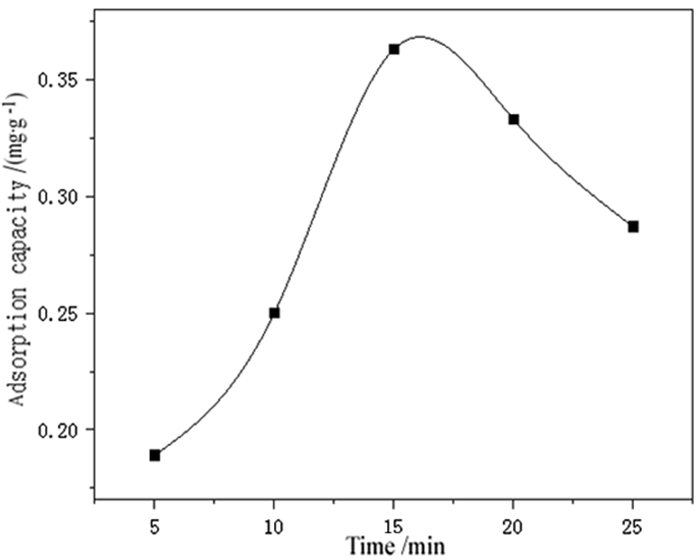
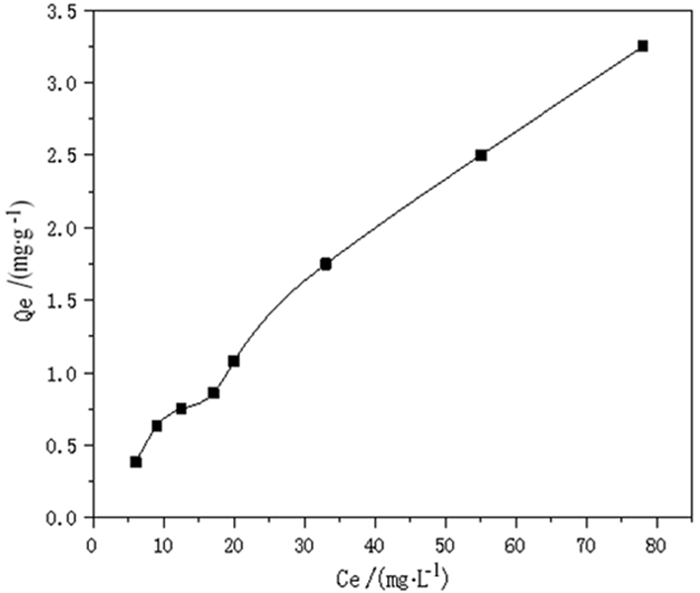
为了强化改性粉煤灰在重金属废水处理中的吸附效果,利用微波联合碱改性的方法,研究微波温度、微波时间、微波功率等制备条件对改性粉煤灰吸附铬(Ⅵ)的影响以及吸附等温特性。结果表明,粉煤灰改性的最佳制备条件为:微波功率600 W,微波温度60℃,微波时间15 min,吸附量达到0.341 mg/g,较改性前提高50%以上。此改性条件下的粉煤灰进行吸附等温的试验研究结果表明,其对铬(Ⅵ)的吸附符合Freundlich和Langmuir等温吸附模型,此吸附过程为单分子层吸附。粉煤灰具有较高的经济性,可广泛用于含铬(Ⅵ)废水的处理。
Abstract:In order to enhance the adsorption effect of modified fly ash in heavy metal wastewater treatment, microwave combined with alkali modification methods was used to study the effects of microwave temperature, microwave time, microwave power and other preparation conditions on the adsorption and the adsorption isotherm characteristic of modified fly ash on Cr(Ⅵ). The results showed that the optimal modified fly ash could be obtained under the conditions of microwave power of 600 W, microwave temperature of 60℃, microwave time of 15 min. The adsorption amount reached 0.341 mg/g, which was more than 50% higher than before modification. The experimental results of adsorption isothermal adsorption of fly ash under this modified condition showed that the adsorption of Cr(Ⅵ)accorded with the isotherm adsorption model of Freundlich and Langmuir, and the adsorption process was monolayer adsorption. Fly ash is highly economical and can be widely used in the treatment of wastewater containing Cr(Ⅵ).
-
Key words:
- microwave-assisted alkali fly ash /
- Cr(Ⅵ) /
- adsorption /
- preparation /
- sorbents
-

-
表 1 粉煤灰的化学成分
Table 1. Chemical composition of fly ash
化学成分 SiO2 Fe2O3 Al2O3 CaO TiO2 K2O MgO BaO MnO Others 含量/% 54.10 17.50 11.60 11.30 2.69 1.18 0.19 0.16 0.13 1.15 表 2 Freundlich和Langmuir等温吸附模型相关参数
Table 2. Parameters of Freundlich and Langmuir isothermal adsorption model
Freundlich isotherm
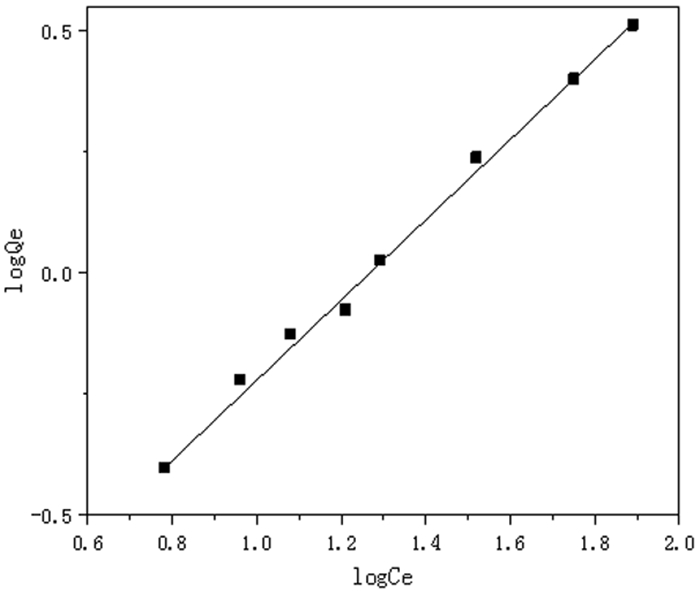
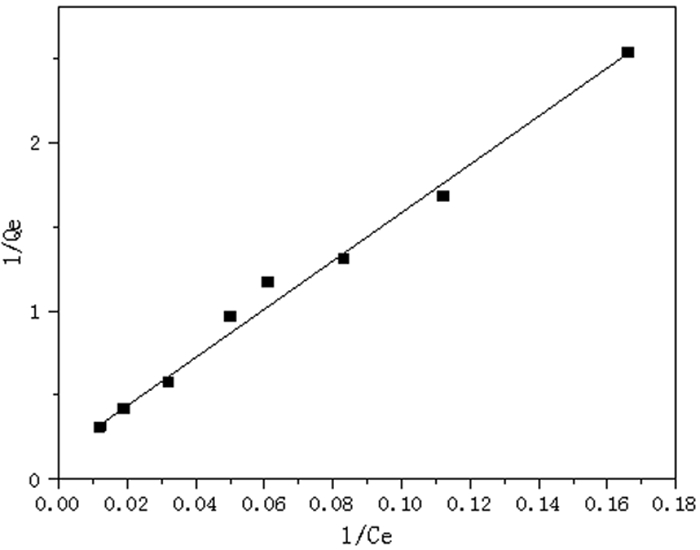
log Qe=0.813 0 log Ce-1.020 0Langmuir isotherm
1/Qe=15.368 4/Ce+0.043 6K 1/n R Xm b R 0.095 5 0.813 0 0.993 0 5.976 2 0.117 7 0.988 8 表 3 不同吸附剂对铬(Ⅵ)的吸附量
Table 3. The adsorption amount of Cr(Ⅵ) with different adsorbent
吸附剂 比表面积/(m2·g-1) 吸附量/(mg·g-1) 未改性粉煤灰 6.104 0.225 改性粉煤灰 20.185 0.341 沸石 600 2.4 -
[1] 李少辉, 赵澜, 包先成, 等.粉煤灰的特性及其资源化综合利用[J].混凝土, 2010(4):76-78. doi: 10.3969/j.issn.1002-3550.2010.04.025
[2] 石大鑫.粉煤灰的综合利用[J].矿产保护与利用, 1992(3):52-53. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=7a28b310-d3b6-4128-ae73-e3bcd6e02f6f
[3] Blissett R S, Rowson N A. A review of the multi-component utilisation of coal fly ash[J]. Fuel, 2012, 97(7):1-23. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=61985f0c6c1d89f9d6cbc53e5d5710bd
[4] Wang S, Wu H. Environmental-benign utilization of fly ash as low-cost adsorbents[J]. J Hazard Mater, 2006, 136(3):482-501. doi: 10.1016/j.jhazmat.2006.01.067
[5] Scheetz B E, Earle R. Utilization of fly ash[J]. Curr opin solid St M, 1998, 3(5):510-520. doi: 10.1016/S1359-0286(98)80017-X
[6] 肖翠微.粉煤灰在水处理领域的应用进展[J].洁净煤技术, 2016, 22(4):45-51. http://d.old.wanfangdata.com.cn/Periodical/jjmjs201604011
[7] 黄琴, 吉伟英, 陈端伟.改性粉煤灰在废水处理中的应用进展[J].上海应用技术学院学报(自然科学版), 2008, 8(1):71-75. doi: 10.3969/j.issn.1671-7333.2008.01.017
[8] Iyer, R. S, Scott, et al. Power station fly ash-a review of value-added utilization outside of the construction industry[J]. Fuel & energy abstra, 2001, 31(3):217-228. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=f64145ac014a6ef4b5e32e3a504d1f4c
[9] 王占华, 周兵, 孙雪景, 等.粉煤灰改性及其在废水处理中的应用现状研究[J].能源环境保护, 2014, 28(4):1-5. http://d.old.wanfangdata.com.cn/Periodical/mkhjbh201404001
[10] 张凡, 欧阳平, 张贤明, 等.粉煤灰改性及其吸附应用研究进展[J].应用化工, 2016, 45(4):747-750. http://d.old.wanfangdata.com.cn/Periodical/sxhg201604036
[11] 于谦.天然高钙粉煤灰的特性及其应用研究[J].矿产保护与利用, 1997, 56(3):48-52. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=a899b0f5-0d06-454c-b7a9-49fae7c5e19c
[12] 李超, 王丽萍, 郭昭华, 等.粉煤灰提铝后尾渣合成13X分子筛及其对Pb2+吸附性能的研究[J].矿产保护与利用, 2018, 218(6):104-108. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=c05b7125-c562-4ef3-8474-92d60d12eecb
[13] 范剑明, 吉仁塔布, 李诚诚, 等.聚硅酸铝铁絮凝剂的制备及其性能研究[J].矿产保护与利用, 2012(4):37-41 doi: 10.3969/j.issn.1001-0076.2012.04.010 http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=d228ad38-951d-4ea4-95df-8b942564aa48
[14] Keng P S, Lee S L, Ha S T, et al. Removal of hazardous heavy metals from aqueous environment by low-cost adsorption materials[J]. Environ chem lett, 2014, 12(1):15-25. doi: 10.1007/s10311-013-0427-1
[15] 胡友彪, 张慰, 王世超.粉煤灰对重金属废水的吸附性能研究[J].煤炭科学技术, 2007, 35(7):96-98. http://d.old.wanfangdata.com.cn/Periodical/mtkxjs200707028
[16] Cho H, Oh D, Kim K. A study on removal characteristics of heavy metals from aqueous solution by fly ash[J]. J Hazard Mater, 2005, 127(1-3):187-195. doi: 10.1016/j.jhazmat.2005.07.019
[17] 彭荣华, 陈丽娟, 李晓湘.改性粉煤灰吸附处理含重金属离子废水的研究[J].材料保护, 2005, 38(1):48-50. doi: 10.3969/j.issn.1001-1560.2005.01.014
[18] 韩晓晶, 张卓, 高淑娟, 等.粉煤灰的改性及在水处理中的研究进展[J].山东化工, 2016, 45(6):43-45. doi: 10.3969/j.issn.1008-021X.2016.06.015
[19] 张凡, 欧阳平, 张贤明, 等.粉煤灰改性及其吸附应用研究进展[J].应用化工, 2016, 45(4):747-750. http://d.old.wanfangdata.com.cn/Periodical/sxhg201604036
[20] 石建稳, 陈少华, 王淑梅, 等.粉煤灰改性及其在水处理中的应用进展[J].化工进展, 2008(3):326-334, 347. doi: 10.3321/j.issn:1000-6613.2008.03.003
[21] Wang Jianlong.Removal of Cr(Ⅵ) from aqueous solution by coal flyash adsorption Ⅰ:Characteristics of chromium adsorption on flyash[J]. Toxicol environ chem, 1999, 68(1):53-62.
[22] Wang Jianlong.Removal of Cr(Ⅵ) from aqueous solution by coal flyash adsorption Ⅱ:Kinetics of chromium adsorption on flyash[J]. Toxicol environ chem, 1999, 68(1-2):63-69. doi: 10.1080/02772249909358645
[23] G. P. Dasmahapatra, T. K. Pal, A. K. Bhadra, et al. Studies on separation characteristics of hexavalent chromium from aqueous solution by fly ash[J]. Sep Sci technol, 1996, 31(14):2001-2009. doi: 10.1080/01496399608001027
[24] 张小庆, 王文洲, 王卫.含铬废水的处理方法[J].环境科学与技术, 2004, 27(1):111-113. http://d.old.wanfangdata.com.cn/Periodical/hjkxyjs2004z1045
[25] 闫旭, 李亚峰.含铬废水的处理方法[J].辽宁化工, 2010, 39(2):143-146. doi: 10.3969/j.issn.1004-0935.2010.02.011
[26] 欧阳平, 张凡, 张贤明, 等.微波辅助改性材料的研究进展[J].应用化工, 2016, 45(1):156-158. http://d.old.wanfangdata.com.cn/Periodical/sxhg201601039
[27] 黄卡玛, 杨晓庆.微波加快化学反应中非热效应研究的新进展[J].自然科学进展, 2006, 16(3):273-279. doi: 10.3321/j.issn:1002-008X.2006.03.004
[28] 唐学红, 肖先举.微波-碱协同改性粉煤灰处理含镉废水的研究[J].化工技术与开发, 2018, 47(9):51-53. http://d.old.wanfangdata.com.cn/Periodical/gxhg201809014
[29] 段小月, 曹红丽, 刘伟, 等.粉煤灰的改性及对刚果红的吸附[J].化工环保, 2013, 33(4):294-298. doi: 10.3969/j.issn.1006-1878.2013.04.003
[30] 徐姝颖, 陈华梅, 贾云雪.氢氧化钠改性粉煤灰处理含铬废水的研究[J].精细石油化工进展, 2011, 12(4):35-37. doi: 10.3969/j.issn.1009-8348.2011.04.009
[31] 郭永龙, 王焰新, 蔡鹤生, 等.水热条件下利用微波加热从粉煤灰合成沸石研究[J].地球科学, 2003(5):517-521. doi: 10.3321/j.issn:1000-2383.2003.05.008
[32] Langmuir I. The constitution and fundamental properties of solids and liquids[J]. J Am Chem Soc, 1916, 38(11):2267. http://d.old.wanfangdata.com.cn/NSTLQK/10.1021-ja02254a006/
[33] Bellot J C, Condoret J S. Modelling of liquid chromatography equilibria[J]. Process biochem, 1993, 28(2):365-376.
-




 下载:
下载: