Comprehensive Recovery of Lithium, Tantalum and Feldspar from Granite Pegmatite Spodumene Ore in Yichun of Jiangxi
-
摘要:
为实现江西宜春花岗伟晶岩型锂辉石矿中锂、钽及长石的综合回收, 开展了选矿综合回收试验研究。研究结果表明, 该锂辉石矿石英、长石含量高, 采用高选择性药剂ZH与氧化石蜡皂组合作为锂辉石捕收剂, 可降低细泥在锂辉石表面的罩盖影响, 优化矿浆流体环境; 在原矿含Li2O为1.51%、Ta2O5为0.022%的条件下, 以氧化石蜡皂+ZH组合捕收剂浮选回收锂辉石, 采用细泥摇床重选工艺回收浮选尾矿中的钽矿物, 重选尾矿采用"弱磁选—强磁选"工艺除铁后作为长石精矿, 获得了含Li2O 5.62%、回收率为74.65%的锂辉石精矿和Ta2O5品位为18.78%、回收率为40.21%的钽精矿, 以及产率为49.16%、含Na2O 2.45%、K2O 4.60%、TFe 0.15%、白度为62.9%的长石精矿。该工艺流程选矿试验指标良好, 实现了硬岩型锂辉石矿中锂、钽和长石的综合回收。
Abstract:In order to realize the efficient comprehensive recovery of lithium, tantalum and feldspar minerals from granite pegmatite type spodumene deposit in Yichun of Jiangxi, the comprehensive recovery test of beneficiation was carried out. The results showed that the combined collector composed of high selectivity reagent ZH and oxidized paraffin soap could realize the effective separation of spodumene with silicate gangue minerals and reduce the influence of fine mud on the mineral surface. Under the condition that the raw ore containing 1.51% Li2O and 0.022% Ta2O5, the comprehensive separation process was as follows: the combined collectors oxidized paraffin soap +ZH were used to flotation spodumene; the tantalum in flotation tailing was recovered by gravity separation process of shaker; and the feldspar was recovered by the separation process of low intensity magnetic separation-high intensity magnetic separation. The process could be obtained spodumene concentrate containing 5.62% of Li2O and recovery rate of 74.65%, tantalum concentrate containing 18.78% of Ta2O5 and recovery rate of 40.21%, feldspar concentrate with yield of 49.16%, Na2O 2.45%, K2O 4.60%, TFe 0.15% and whiteness of 62.9%. The beneficiation test index of the process was good, which realized the comprehensive recovery of lithium, tantalum and feldspar from hard rock spodumene ore.
-
Key words:
- spodumene /
- tantalum /
- feldspar /
- collector /
- comprehensive recovery /
- flotation separation /
- gravity separation /
- magnetic separation
-

-
表 1 试样化学多元素分析结果
Table 1. Chemical multi-element analysis results of samples
/% 元素 Li2O Ta2O5 Nb2O5 TFe SiO2 P 含量 1.51 0.0223 0.0051 1.96 70.50 0.046 元素 Al2O3 CaO MgO Na2O K2O 含量 13.76 0.22 0.23 3.80 1.93 表 2 矿物含量分析结果
Table 2. Analysis results of mineral content
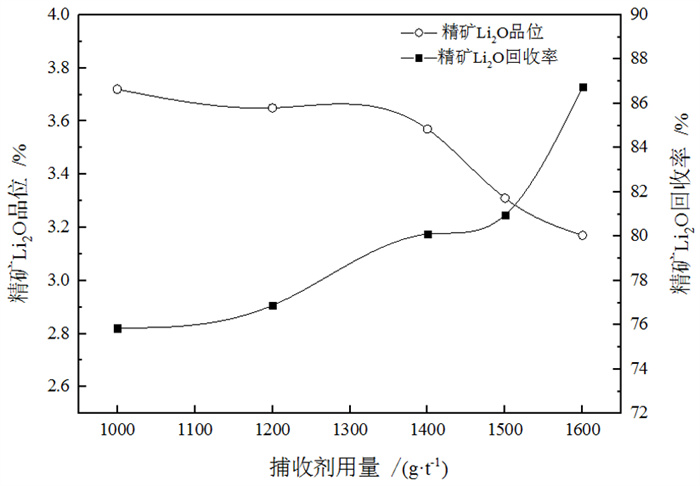
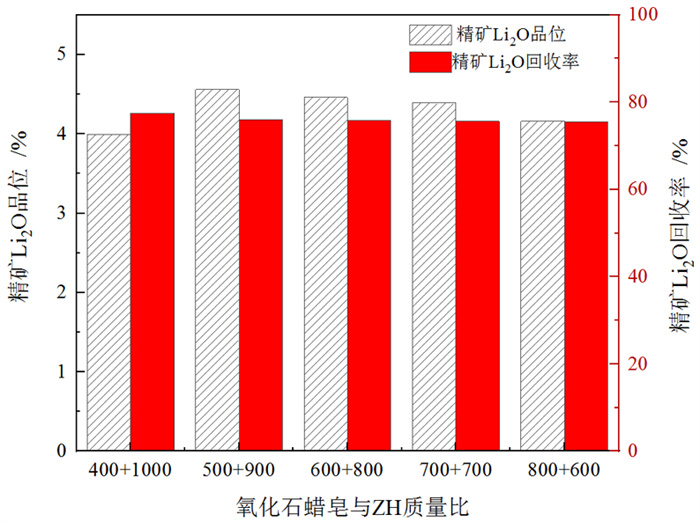
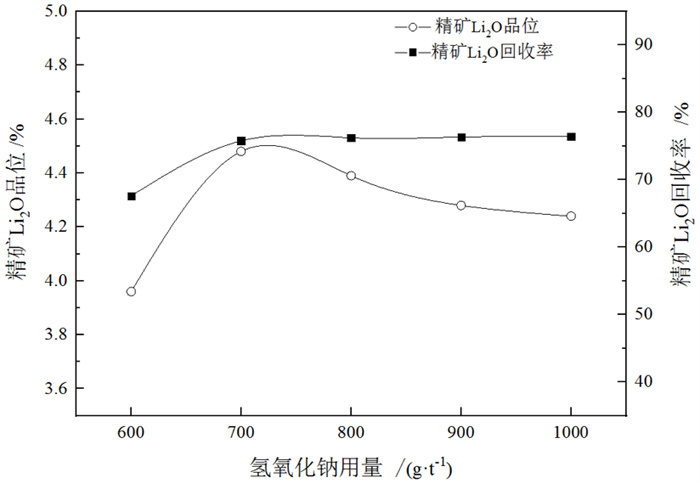
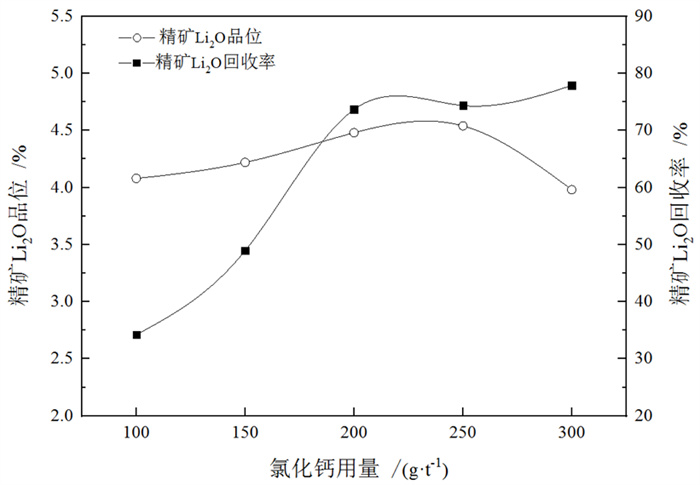
/% 矿物 钾长石 斜长石 石英 锂辉石 磷灰石 白云母 钠长石 含量 13 20 35 15 1 8 8 表 3 捕收剂种类试验结果
Table 3. Test results of collector type
/% 捕收剂种类 产率 Li2O品位 Li2O回收率 氧化石蜡皂 24.78 3.57 58.34 植物油酸 17.86 4.13 51.76 ZH 18.31 4.78 57.26 氧化石蜡皂+油酸 34.26 3.57 80.52 ZH+氧化石蜡皂 23.81 4.39 77.87 表 4 钽重选试验结果
Table 4. Tantalum gravity separation test results
/% 产品名称 产率 Ta2O5品位 Ta2O5回收率 钽精矿 0.04 18.43 40.24 尾矿 99.96 0.01 59.76 浮选尾矿 100.00 0.02 100.00 表 5 强磁选场强条件试验结果
Table 5. Test results of strong magnetic separation field strength conditions
/% 磁场场强 产品名称 产率 TFe品位 TFe回收率 1.0 T+1.3 T 弱磁选铁杂质 20.33 2.89 68.33 强磁粗选铁杂质 11.50 1.17 15.65 强磁精选铁杂质 7.25 0.64 5.40 长石粉 60.92 0.15 10.63 重选尾矿 100.00 0.86 100.00 1.0 T+1.5 T 弱磁选铁杂质 20.17 2.94 68.94 强磁粗选铁杂质 11.75 1.12 15.30 强磁精选铁杂质 8.08 0.71 6.69 长石粉 60.00 0.13 9.07 重选尾矿 100.00 0.86 100.00 表 6 长石精矿多元素分析
Table 6. Multi element analysis of feldspar concentrate
/% 元素 Li2O TFe SiO2 Al2O3 CaO MgO Na2O K2O 含量 0.11 0.15 76.9 12.60 0.06 0.0085 4.60 2.45 表 7 全流程工艺试验结果
Table 7. Test results of the whole process
/% 产品名称 产率 品位 回收率 Li2O Ta2O5 TFe Na2O K2O Li2O Ta2O5 TFe Na2O K2O 锂辉石精矿 20.06 5.62 0.039 0.02 / / 74.65 35.25 0.20 / / 钽精矿 0.05 0.53 18.78 4.36 / / 0.02 40.21 0.11 / / 长石精矿 49.16 0.11 0.008 0.15 4.60 2.45 3.58 17.88 3.76 59.51 62.41 杂质矿物 30.73 1.07 0.005 6.12 / / 21.75 6.66 95.93 / / 原矿 100.00 1.51 0.022 1.96 3.80 1.93 100.00 100.00 100.00 100.00 100.00 -
[1] ZHANG Y B, ZHOU H P, CAO Y J, et al. Activation mechanism of calcium hydrolysate on the spodumene surface and its effect on the adsorption of collector[J]. Minerals Engineering, 2021, 174: 107221. doi: 10.1016/j.mineng.2021.107221
[2] ZHOU H P, HU J, ZHANG Y B, et al. Effectively enhancing recovery of fine spodumene via aggregation flotation[J]. Rare metals, 2020, 39(3): 316-326. doi: 10.1007/s12598-019-01365-5
[3] SHU K Q, XU L H, WU H Q, et al. In situ adsorption of mixed collectors BHA/DDA in spodumene-feldspar flotation system[J]. Separation and Purification Technology, 2020, 251: 117325. doi: 10.1016/j.seppur.2020.117325
[4] YANG Y, OKONKWO E G, HUANG G Y, et al. On the sustainability of lithium ion battery industry-a review and perspective[J]. Energy Storage Materials, 2021, 36: 186-212. doi: 10.1016/j.ensm.2020.12.019
[5] QIAO D H, WANG G S, GAO T M, et al. Potential impact of the end-of-life batteries recycling of electric vehicles on lithium demand in China: 2010—2050[J]. Science of the Total Environment, 2021, 764: 142835. doi: 10.1016/j.scitotenv.2020.142835
[6] 孙传尧, 印万忠. 硅酸盐矿物浮选原理[M]. 北京: 科学出版社, 2001: 231-234.
SUN C Y, YIN W Z. Flotation principle of silicate minerals[M]. Beijing: Science Press, 2001: 231-234.
[7] 杨卉芃, 柳林, 丁国峰. 全球锂矿资源现状及发展趋势[J]. 矿产保护与利用, 2019, 39(5): 26-40. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=f8716804-8b43-4b0f-8070-72884ef8cc74
YANG H P, LIU L, DING G F. Present situation and development trend of lithium resources in the world[J]. Conservation and Utilization of Mineral Resources, 2019, 39(5): 26-40. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=f8716804-8b43-4b0f-8070-72884ef8cc74
[8] 周贺鹏, 张永兵, 雷梅芬, 等. 磁选尾矿综合回收钽铌锂及长石选矿工艺研究[J]. 非金属矿, 2018, 41(3): 69-71. doi: 10.3969/j.issn.1000-8098.2018.03.023
ZHOU H P, ZHANG Y B, LEI M F, et al. Study on the comprehensive recovery of tantalum and niobium lithium and feldspar from magnetic separation tailings[J]. Non-Metallic Mines, 2018, 41(3): 69-71. doi: 10.3969/j.issn.1000-8098.2018.03.023
[9] 张垒, 王毓华, 于福顺, 等. 四川某低品位难选锂辉石矿选矿工艺研究[J]. 有色金属(选矿部分), 2015(6): 50-53. doi: 10.3969/j.issn.1671-9492.2015.06.012
ZHANG L, WANG Y H, YU F S, et al. Research on mineral processing technology for a low grade spodumene ore in Sichuan[J]. Nonferrous Metals(Mineral Processing Section), 2015(6): 50-53. doi: 10.3969/j.issn.1671-9492.2015.06.012
[10] 吕永信, 幸伟中, 李金荣. 概论锂辉石和绿柱石矿石浮选理论与实践[J]. 有色金属(冶炼部分), 1965(6): 14-19+25. https://www.cnki.com.cn/Article/CJFDTOTAL-METE196506003.htm
LV Z X, XIN W Z, LI J R. Introduction to flotation theory and practice of spodumene and beryl ores[J]. Nonferrous Metals(Extractive Metallurgy), 1965(6): 14-19+25. https://www.cnki.com.cn/Article/CJFDTOTAL-METE196506003.htm
[11] 麦笑宇, 梅光军, 余永富. 有机酸抑制剂对含铁硅酸盐矿物的抑制机理研究[J]. 矿冶工程, 2001, 21(4): 25-28. doi: 10.3969/j.issn.0253-6099.2001.04.009
MAI X Y, MEI G J, YU Y F. Depressing mechanism of organic acid depressors on ferrosilicate minerals study[J]. Mining and Metallurgical Engineering, 2001, 21(4): 25-28. doi: 10.3969/j.issn.0253-6099.2001.04.009
[12] 钱志博, 周少珍. 某低品位锂辉石矿石浮选试验研究[J]. 有色金属(选矿部分), 2019(6): 62-68. doi: 10.3969/j.issn.1671-9492.2019.06.012
QIAN Z B, ZHOU S Z. Experimental study on flotation of a low grade spodumene ore[J]. Nonferrous Metals(Mineral Processing Section), 2019(6): 62-68. doi: 10.3969/j.issn.1671-9492.2019.06.012
[13] 朱一民, 谢瑞琦, 张猛. 锂辉石浮选捕收剂及调整剂研究综述[J]. 金属矿山, 2019(2): 15-21. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS201902005.htm
ZHU Y M, XIE R Q, ZHANG M. Review on research of spodumene flotation collector and regulator[J]. Metal Mine, 2019(2): 15-21. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS201902005.htm
[14] 吕永信. 锂辉石—绿柱石浮选分离新方法——污染离子Ca2+选择性解吸分离法[J]. 矿产综合利用, 1980(1): 8-16. https://www.cnki.com.cn/Article/CJFDTOTAL-KCZL198001001.htm
LV Y X. A new flotation separation method of spodumene beryl—selective desorption separation of polluted ion Ca2+[J]. Multipurpose Utilization of Mineral Resources, 1980(1): 8-16. https://www.cnki.com.cn/Article/CJFDTOTAL-KCZL198001001.htm
[15] 王毓华, 于福顺. 新型捕收剂浮选锂辉石和绿柱石[J]. 中南大学学报(自然科学版), 2005, 36(5): 93-97. https://www.cnki.com.cn/Article/CJFDTOTAL-ZNGD200505017.htm
WANG Y H, YU F S. Flotation of spodumene and beryl with a new collector[J]. Journal of Central South University(Science and Technology), 2005, 36(5): 93-97. https://www.cnki.com.cn/Article/CJFDTOTAL-ZNGD200505017.htm
[16] 李云, 李茂林, 崔瑞, 等. 某伟晶岩型锂辉石矿石浮选试验[J]. 金属矿山, 2019(1): 87-91. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS201901017.htm
LI Y, LI M L, CUI R, et al. Flotation test of a pegmatite spodumene ore[J]. Metal Mine, 2019(1): 87-91. https://www.cnki.com.cn/Article/CJFDTOTAL-JSKS201901017.htm
[17] HE G, XIANG H, JIANG W, et al. First-principles theory on electronic structure and floatability of spodumene[J]. Rare Metals, 2014, 33(6): 742-748. doi: 10.1007/s12598-014-0317-0
[18] 周贺鹏. 微细粒锂辉石聚团浮选特性及矿物表面反应机理[D]. 徐州: 中国矿业大学, 2020.
ZHOU H P. Flotation characteristics and mineral surface reaction mechanism of fine spodumene agglomerates[D]. Xuzhou: China university of mining and technology, 2020.
[19] TIAN J, XU L, DENG W, et al. Adsorption mechanism of new mixed anionic/cationic collectors in a spodumene-feldspar flotation system[J]. Chemical Engineering Science, 2017, 164: 99-107.
[20] 李金林, 刘丹, 王伊杰, 等. 锂辉石浮选机理研究现状[J]. 硅酸盐通报, 2016, 35(8): 2400-2406+2411. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT201608013.htm
LI J L, LIU D, WANG Y J, et al. Research status of spodumene flotation mechanism[J]. Bulletin of the Chinese Ceramic Society, 2016, 35(8): 2400-2406+2411. https://www.cnki.com.cn/Article/CJFDTOTAL-GSYT201608013.htm
[21] WANG Y, ZHU G, LEI Z, et al. Surface dissolution of spodumene and its role in the flotation concentration of a spodumene ore[J]. Minerals Engineering, 2018, 125: 120-125.
[22] 张永兵. 油酸钠体系下锂辉石浮选过程的量子化学计算[D]. 赣州: 江西理工大学, 2020.
ZHANG Y B. Quantum chemical calculation of spodumene flotation process in sodium oleate system[D]. Ganzhou: Jiangxi university of science and technology, 2020.
[23] 胡阳, 吴港生, 褚浩然, 等. 锂辉石矿浮选理论与药剂研究的新进展[J]. 有色金属工程, 2021, 11(11): 10-19. https://www.cnki.com.cn/Article/CJFDTOTAL-YOUS202111003.htm
HU Y, WU G S, CHU H R, et al. New progress in flotation theory and reagent research of spodumene ore[J]. Nonferrous Metal Engineering, 2021, 11(11): 10-19. https://www.cnki.com.cn/Article/CJFDTOTAL-YOUS202111003.htm
[24] 谢贞付, 王毓华, 于福顺, 等. 伟晶岩型锂辉石矿浮选研究综述[J]. 稀有金属, 2013, 37(4): 641-649. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXJS201304020.htm
XIE Z F, WANG Y H, YU F S, et al. Reviews of flotation research on pegmatite spodumene ores[J]. Chinese Journal of Rare Metals, 2013, 37(4): 641-649. https://www.cnki.com.cn/Article/CJFDTOTAL-ZXJS201304020.htm
-



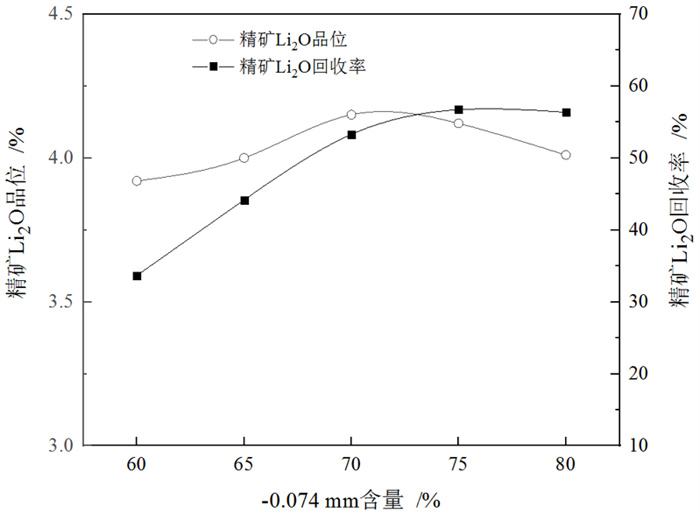
 下载:
下载: