Study on Influence Factors for Determination of Oxygen Isotopic Composition of Silicates and Oxide Minerals by BrF5 Method
-
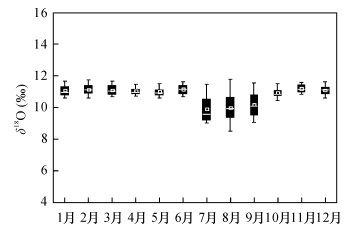
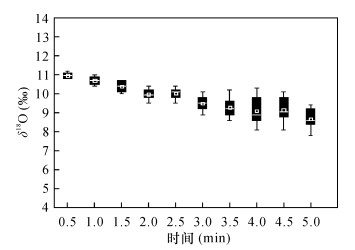
摘要: 应用五氟化溴法(BrF5)测定硅酸盐及氧化物矿物的氧同位素组成,试剂纯化、样品预处理、环境湿度、反应器洁净程度以及实验质量控制是影响该法准确度的主要因素。为确定BrF5法最佳实验条件,提高分析精度,本文以国家标准物质为参考,对硅酸盐及氧化物矿物在不同焙烧温度下的氧同位素组成进行分析,并对实验环境湿度和反应器洁净程度对分析数据质量的影响进行了系统研究。实验确定了焙烧温度设置为650~750℃,可去除硅酸盐全岩样品中的炭质成分,避免了温度因素引起的同位素分馏;将实验环境相对湿度降低至50% RH,并在0.5min内完成样品装填,可解决由于水汽造成δ18O值明显偏低的突出问题;对于新使用的反应器,或反应器在使用中有黄色物质脱落、内壁受到污染,及时进行氟化处理和清洗,可避免因氧气产率降低所引起的氧同位素分馏;反应器交替进行样品和标准物质CO2试样的制备,可对样品同位素数据的可靠性进行监测。对国家标准GBW04409的氧同位素组成进行测试,δ18O测试结果与推荐值一致,分析精度优于0.1‰。以上途径有效控制了氧同位素分馏,提高了BrF5氧同位素分析方法的适用性。Abstract:
BACKGROUNDReagent purification, sample pretreatment, environmental humidity, reactor cleanliness and experimental quality control are the main influence factors for determination of oxygen isotopic composition of silicates and oxide minerals by BrF5 method. OBJECTIVESTo determine the optimal experimental conditions and improve the accuracy by selecting silicates and oxide minerals as laboratory raw materials for oxygen isotopic composition analysis. To discuss the influence factors for determination of oxygen isotopic composition of silicates and oxide minerals by BrF5 method. METHODSUsing BrF5 analysis method and taking standard sample as the reference, oxygen isotopic compositions of silicates and oxide minerals were determined at different calcination temperatures. At the same time, the effects of environmental humidity and reactor cleanliness on the analytical results were also studied. RESULTSThe results show that 650-750℃ is the best calcination temperature range for removing carbon components during oxygen isotope analysis, which can avoid the isotope fractionation caused by temperature. By reducing the relative environment humidity or the sample loading time within 0.5min, the lower δ18O values caused by vapor was improved. The new reactor, or the reactor with large yellow substance falling off, or the contaminated reactor will reduce the yield of oxygen and cause oxygen isotope fractionation, so the reactors should be fluorinated or purged in time. The sample and standard material is loaded alternately for each reactor to monitor the reliability of the isotopic composition of the sample. The results of oxygen isotopic composition of GBW04409 are consistent with the recommended values. The relative standard deviation is better than 0.1‰ for δ18O under the optimized conditions. CONCLUSIONSThe proposed method can effectively control oxygen isotope fractionation and improve the applicability of BrF5 oxygen isotope analysis method. -

-
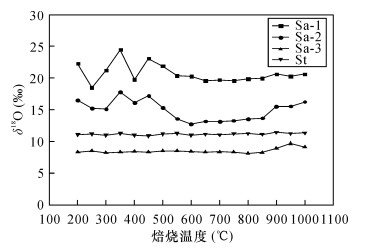
表 1 在不同焙烧温度下测定样品的有机碳含量及颜色变化情况
Table 1. Measured organic carbon content in samples and their color information at different calcination temperature
焙烧温度
(℃)黑色含炭质硅质页岩
(Sa-1)黑色含炭质黏土质页岩
(Sa-2)有机碳含量
(%)样品颜色 有机碳含量
(%)样品颜色 100 4.3825 黑色 2.1145 黑色 150 4.3765 黑色 2.1142 黑色 200 4.3124 黑色 2.0116 黑色 250 4.2939 黑色 1.9427 浅黑色 300 4.2276 浅黑色 1.8739 浅黑色 350 3.0262 浅黑色 1.4662 浅灰色 400 1.5870 浅灰黑色 0.2394 浅灰色 450 0.6530 浅灰色 0.1695 灰白色 500 0.0314 浅灰色 0.0374 灰白色 550 0.0284 灰白色 0.0251 灰白色 600 0.0211 灰白色 0.0088 灰白色 650 0.0090 灰白色 0.0076 灰白色 700 0.0024 灰白色 0.0053 灰白色 750 0.0021 灰白色 0.0035 灰白色 800 0.0016 灰白色 0.0015 灰白色 850 0.0011 灰白色 0.0012 灰白色 900 0.0008 灰白色 0.0009 灰白色 950 0.0008 灰白色 0.0008 灰白色 1000 0.0006 灰白色 0.0008 灰白色 -
[1] Taylor H P.The oxygen isotope geochemistry of igneous rocks[J].Contribution to Mineralogy and Petrology, 1968, 19:1-71. doi: 10.1007/BF00371729
[2] 李铁军.氧同位素在岩石成因研究的新进展[J].岩矿测试, 2013, 32(6):841-849. doi: 10.3969/j.issn.0254-5357.2013.06.002 http://www.ykcs.ac.cn/article/id/19705ed2-36ac-44a8-82a4-0a8cd5251392
Li T J.Progress in the application of oxygen isotopes in the study of petrogenesis[J].Rock and Mineral Analysis, 2013, 32(6):841-849. doi: 10.3969/j.issn.0254-5357.2013.06.002 http://www.ykcs.ac.cn/article/id/19705ed2-36ac-44a8-82a4-0a8cd5251392
[3] Clayton R N, Mayeda T K.The use of bromine pentafluo-ride in the extraction of oxygen from oxides and silicates for isotopic analysis[J].Geochimica et Cosmochimica Acta, 1963, 27:43-52. doi: 10.1016/0016-7037(63)90071-1
[4] Clayton R N.High Temperature Isotope Effects in the Early Solar System[M]//Valley J W, eds.Stable Isotopes in High Temperature Geological Processes.Review in Mineralogy and Geochemistry, 1986, 16: 129-139.
[5] Clayton R N, Mayeda T K, Epstein S.Isotopic Fractiona-tion of Silicon in Allende Inclusions[C]//Proceedings of the 9th Lunar and Planetary Science Conference.Houston, 1978: 1267-1278.
http://adsabs.harvard.edu/abs/1978LPSC....9.1267C [6] 丁悌平, 万德芳, 李金城, 等.硅同位素测量方法及其地质应用[J].矿床地质, 1988, 7(4):90-95. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK000001933966
Ding T P, Wan D F, Li J C, et al.The analytic method of silicon isotopes and its geological application[J].Mineral Deposits, 1988, 7(4):90-95. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK000001933966
[7] 李延河, 万德芳, 张国柄, 等.氧化物、硅酸盐矿物的氧同位素分析方法——BrF5法[M]//稳定同位素分析方法研究进展.北京:北京科学技术出版社, 1992:37-43.
Li Y H, Wan D F, Zhang G B, et al.A Study on BrF5Method of Oxygen Isotope Analyses of Oxides and Silicates[M]//Progress of Analytic Methods of Stable Isotopes.Beijing:Beijing Science and Technology Publishing House, 1992:37-43.
[8] 万德芳, 李延河.硫酸盐的氧同位素测量方法[J].高校地质学报, 2006, 12(3):378-383. doi: 10.3969/j.issn.1006-7493.2006.03.010
Wan D F, Li Y H.Analytical method of oxygen isotope composition in sulphates[J].Geological Journal of China Universitys, 2006, 12(3):378-383. doi: 10.3969/j.issn.1006-7493.2006.03.010
[9] 秦燕, 李延河, 刘锋, 等.新疆吐-哈地区硝酸盐矿床的氧同位素非质量效应[J].地球学报, 2008, 29(6):729-734. doi: 10.3321/j.issn:1006-3021.2008.06.011
Qin Y, Li Y H, Liu F, et al.Mass independent oxygen isotope fractionation in nitrate deposits of the Turpan-Hami area, Xinjiang[J].Acta Geoscientica Sinica, 2008, 29(6):729-734. doi: 10.3321/j.issn:1006-3021.2008.06.011
[10] Sharp Z D.A laser-based microanalytical method for the in situ determination of oxygen isotope ratios of silicates and oxides[J].Geochimica et Cosmochimica Acta, 1990, 54(5):1353-1357. doi: 10.1016/0016-7037(90)90160-M
[11] Sharp Z D.In situ laser microprobe techniques for stable isotope analysis[J].Chemical Geology (Isotope Geoscience Section), 1992, 101:3-19. doi: 10.1016/0009-2541(92)90199-F
[12] 肖益林, 傅斌, 郑永飞.激光探针分析在氧同位素地球化学研究中的应用[J].地学前缘, 1998, 5(1-2):283-294. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199800062468
Xiao Y L, Fu B, Zheng Y F.Applications of laserprobe method to oxygen isotope geochemistry[J].Earth Science Frontiers, 1998, 5(1-2):283-294. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199800062468
[13] 龚冰, 郑永飞, 赵子福, 等.硅酸盐和金属氧化物矿物氧同位素组成的CO2激光氟化分析[J].矿物岩石地球化学通报, 2001, 20(4):428-430. doi: 10.3969/j.issn.1007-2802.2001.04.063
Gong B, Zheng Y F, Zhao Z F, et al.Oxygen isotope analysis of silicate and metal oxide minerals by CO2 laser fluorination[J].Bulletin of Mineralogy, Petrology and Geochemistry, 2001, 20(4):428-430. doi: 10.3969/j.issn.1007-2802.2001.04.063
[14] 龚冰, 郑永飞.硅酸盐矿物氧同位素组成的激光分析[J].地学前缘, 2003, 10(2):279-286. doi: 10.3321/j.issn:1005-2321.2003.02.003
Gong B, Zheng Y F.A CO2-laser technique for oxygen isotope analysis of silicates[J].Earth Science Frontiers, 2003, 10(2):279-286. doi: 10.3321/j.issn:1005-2321.2003.02.003
[15] 高建飞, 丁悌平.激光熔蚀微量氧同位素分析方法及其地质应用[J].地质论评, 2007, 54(1):139-144. http://d.old.wanfangdata.com.cn/Periodical/dzlp200801016
Gao J F, Ding T P.Laser microprobe oxygen isotope analysis method and geology applications[J].Geological Review, 2007, 54(1):139-144. http://d.old.wanfangdata.com.cn/Periodical/dzlp200801016
[16] 刘熙, 邓文峰, 魏静娴, 等.利用激光氟化系统分析硅酸盐矿物的三氧同位素组成[J].矿物岩石地球化学通报, 2016, 35(3):448-453. doi: 10.3969/j.issn.1007-2802.2016.03.006
Liu X, Deng W F, Wei J X, et al.Analysis of triple oxygen isotopic compositions of silicate minerals by using laser fluorination system[J].Bulletin of Mineralogy, Petrology and Geochemistry, 2016, 35(3):448-453. doi: 10.3969/j.issn.1007-2802.2016.03.006
[17] Johnson W D.Method and Apparatus for Laser Pest Con-trol[P].United States, 5915949[1994-06-29].
[18] 郑永飞, 陈江峰.稳定同位素地球化学[M].北京:科学出版社, 2000.
Zheng Y F, Chen J F.Stale Isotope Geochemistry[M].Beijing:Science Press, 2000.
[19] Elsenheimer D, Valley J W.In situ oxygen isotope analy-sis of feldspar and quartz by Nd:YAG laser microprobe[J].Chemical Geology:Isotope Geoscience Section, 1992, 101(1-2):21-42. doi: 10.1016/0009-2541(92)90200-O
[20] Wiechert U, Fiebig J.Excimer laser isotope-ratio-monito-ring mass spectrometry for in situ oxygen isotope analysis[J].Chemical Geology, 2002, 182(2-4):179-194. doi: 10.1016/S0009-2541(01)00287-X
[21] Valley J W, Kinny P D, Schulze D J, et al.Zircon mega-crysts from kimberlite:Oxygen isotope variability among mantle melts[J].Contributions to Mineralogy and Petrology, 1998, 133(1-2):1-11. doi: 10.1007/s004100050432
[22] 丁悌平.稳定同位素测试技术与参考物质研究现状及发展前景[J].岩矿测试, 2002, 21(4):291-300. doi: 10.3969/j.issn.0254-5357.2002.04.011 http://www.ykcs.ac.cn/article/id/ykcs_20020477
Ding T P.Present status and prospect of analytical techniques and reference materials for stable isotopes[J].Rock and Mineral analysis, 2002, 21(4):291-300. doi: 10.3969/j.issn.0254-5357.2002.04.011 http://www.ykcs.ac.cn/article/id/ykcs_20020477
[23] 郑淑慧, 郑斯成, 莫志超.稳定同位素地球化学分析[M].北京:北京大学出版社, 1986:194-216.
Zheng S H, Zheng S C, Mo Z C.Stable Isotope Analyses of Geochemistry[M].Beijing:Peking University Press, 1986:194-216.
[24] 石晓, 刘汉彬, 张建锋, 等.BrF5法分析难熔矿物氧同位素组成的反应温度探讨[J].世界核地质科学, 2018, 35(1):52-59. doi: 10.3969/j.issn.1672-0636.2018.01.008
Shi X, Liu H B, Zhang J F, et al.Study on reaction temperature of oxygen isotopes composition of refractory minerals using BrF5method[J].World Nuclear Geoscience, 2018, 35(1):52-59. doi: 10.3969/j.issn.1672-0636.2018.01.008
[25] 袁维玲, 潘飞云.氧同位素分析方法——BrF5法的实验研究[J].矿产与地质, 1996, 10(5):356-361. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199600748597
Yuan W L, Pan F Y.Experiment study of BrF5 method-An analysis method of oxygen isotope[J].Mineral Resources and Geology, 1996, 10(5):356-361. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199600748597
[26] 丁悌平, 黎红, 张国柄, 等.六氟化硫法测量33S/32S、34S/32S和36S/32S的方法研究[J].矿床地质, 1987, 6(4):81-93. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK000004298587
Ding T P, Li H, Zhang G B, et al.A study on determining 33S/32S, 34S/32S and 36S/32S by SF6 method[J].Mineral Deposits, 1987, 6(4):81-93. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK000004298587
[27] 丁悌平, 蒋少涌, 万德芳, 等.硅同位素地球化学[M].北京:地质出版社, 1994:9-12.
Ding T P, Jiang S Y, Wan D F, et al.Silicon Isotope Geochemistry[M].Beijing:Geological Publishing House, 1994:9-12.
-



 下载:
下载: