Particle Size Distribution of Perfluoroalkyl Substances in Atmospheric Particulate Matter in Beijing
-
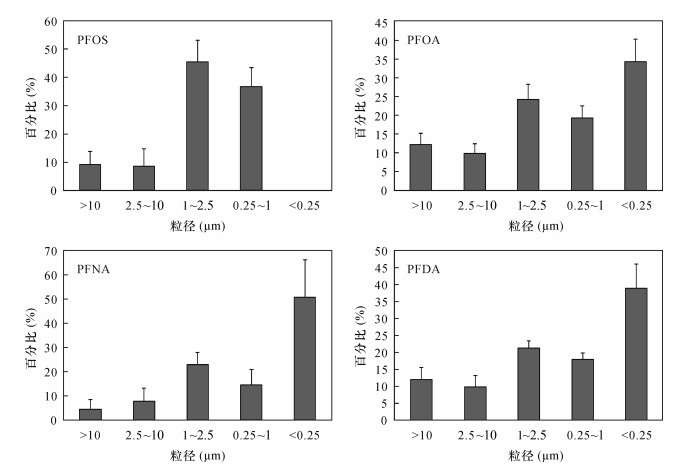
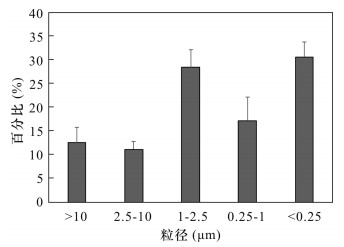
摘要: 人为排放的持久性有机污染物倾向于在细级大气颗粒物中富集,但目前国内外关于大气颗粒物中全氟烷基化合物(PFASs)粒径分布在不同国家地区有显著差异,而在我国北京地区PFASs在不同粒径大气颗粒物中的富集能力尚不清楚。本文采用五级大流量主动分级采样器采集了北京市大气颗粒物样品,利用超声萃取结合高效液相色谱-电喷雾负电离源串联质谱测定PFASs含量,探讨了该地区大气颗粒物中PFASs的浓度水平和粒径分布特征,以及大气颗粒物浓度变化对PFASs浓度变化的影响。结果表明:研究区∑PFASs范围为10.1~62.9 pg/m3,76.4%~83.8%的PFASs集中分布在PM2.5颗粒物中,其中含量较高的PFOA、PFNA和PFDA在<0.25 μm细颗粒物中占比最高,分别为26.3%~43.7%、30.3%~68.6%和30.6%~49.7%;PFOS在<0.25 μm细颗粒物中没有检出,主要分布在1~2.5 μm和0.25~1 μm颗粒物中。此外,研究发现北京市霾天大气颗粒物中∑PFASs为晴天的3.5倍,且不同粒径大气颗粒物浓度变化对PFASs各化合物表现出不同的富集能力,其中PFOA、PFOS、PFNA和PFDA等中链PFASs更易富集。Abstract:
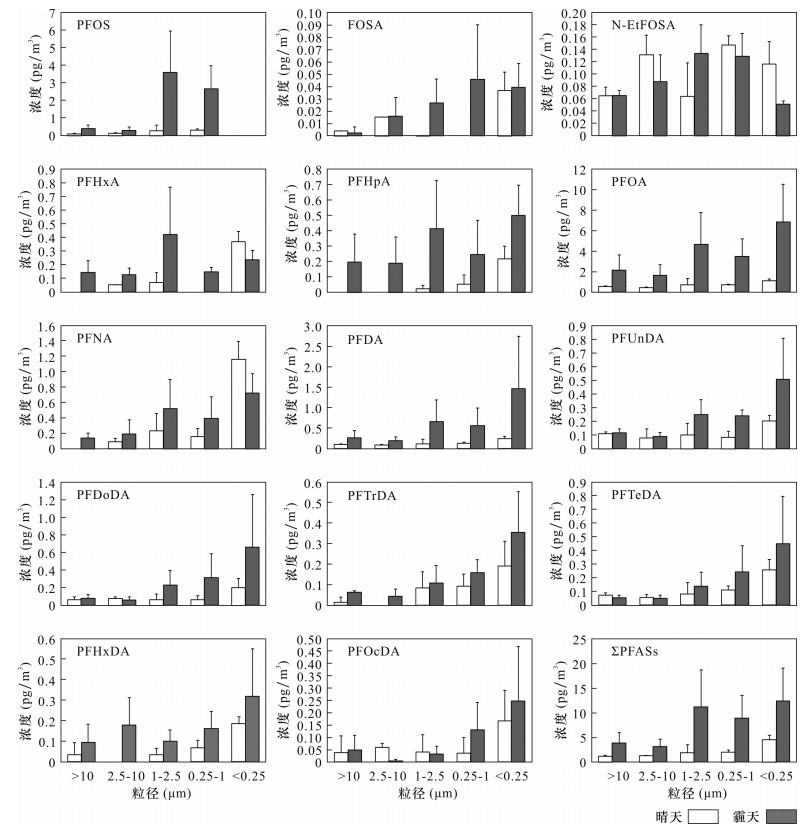
BACKGROUND Anthropogenic emissions of persistent organic pollutants (POPs) tend to be concentrated in fine-level atmospheric particles. However, there are significant differences in the particle size distribution of perfluoroalkyl compounds (PFASs) in atmospheric particulate matter in different countries and regions, and the enrichment capacity of PFASs in different size particulate matter in Beijing is still unclear. OBJECTIVES To reveal the concentration level and particle size distribution characteristics of perfluoroalkyl substances in atmospheric particulate matter in Beijing, by collecting samples using a five stage impactor sampler and to discuss the effects of atmospheric particulate matter concentration changes on the concentration of perfluoroalkyl substances under different sizes. METHODS The samples were pretreated by ultrasonic extraction, and the concentrations of PFASs were determined by High Performance Liquid Chromatography-Electrospray Negative Ionization Source Tandem Mass Spectrometry. RESULTS The concentrations of ∑PFASs are 10.1-62.9 pg/m3, and 76.4%-83.8% of PFASs are concentrated in PM2.5. PFOA, PFNA and PFDA have the highest concentration in the <0.25 μm particles, accounting for 26.3%-43.7%, 30.3%-68.6% and 30.6%-49.7%, respectively. PFOS is not detected in the fine particles (0.25 μm) and 68.9%-2.8% of PFOS are concentrated in the particles with sizes of 0.25-1 μm and 1-2.5 μm. CONCLUSIONS The ∑PFASs in the atmospheric particles of smog in Beijing are 3.5 times that of sunny days, and the changes of atmospheric particle concentration of different particle sizes show different enrichment ability for each compound of PFASs, among which PFOA, PFOS, PFNA and PFDA middle chain PFASs are enriched more easily. -

-
表 1 北京市大气样品采集信息
Table 1. Sampling information in Beijing city
采样日期
(2014年)采样时间
(h)天气
状况室外温度
(℃)空气质量
等级AQI
指数10月9日 24 霾 13~22 严重污染 380 10月13日 24 晴 2~17 优 29 10月16日 24 晴 6~22 良 73 10月18日 24 晴 13~22 中度污染 194 10月21日 24 霾 7~17 重度污染 201 10月23日 24 霾 12~18 严重污染 263 10月31日 24 霾 8~17 良 67 注:AQI指数为空气污染指数,根据环境空气质量标准和各项污染物对人体健康、生态、环境的影响,将常规监测的几种空气污染物浓度简化成为单一的概念性指数值形式,它将空气污染程度和空气质量状况分级表示,适合于表示城市的短期空气质量状况和变化趋势。 表 2 大气样品中各PFASs的方法检出限、平均回收率及加标回收率
Table 2. Detection limits of the method and recoveries for individual PFASs in air samples
PFASs化合物 LOD
(pg/m3)平均回收率±
标准偏差(%)加标回收率±
标准偏差(%)同位素标记化合物 平均回收率±
标准偏差(%)PFHxA 0.005 103.2±11.3 97.5±13.6 13C2PFHxA 117.6±9.8 PFHpA 0.005 98.9±15.6 98.3±16.4 13C4PFOA 106.8±10.4 PFOA 0.005 104.3±12.7 117.5±11.9 13C5PFNA 115.5±15.1 PFNA 0.005 101.1±18.5 100.4±20.3 13C2PFDA 111.7±9.2 PFDA 0.023 99.3±13.8 98.9±14.1 13C2PFUnDA 104.5±12.6 PFUnDA 0.023 89.7±12.5 75.8±10.7 13C2PFDoDA 101.9±14.2 PFDoDA 0.005 102.5±9.8 120.3±12.9 13C4PFOS 121.6±16.3 PFTrDA 0.005 123.1±17.3 103.2±18.9 PFTeDA 0.005 102.6±14.0 87.1±17.4 PFHxDA 0.005 102.5±19.5 100.2±21.6 PFOcDA 0.005 92.6±11.4 107.7±15.1 PFOS 0.005 100.9±16.3 101.8±14.5 FOSA 0.006 93.6±13.2 90.5±12.7 N-EtFOSA 0.005 96.2±12.1 93.5±14.5 表 3 北京市大气颗粒物中PFASs组成
Table 3. Composition of PFASs in atmospheric particulates in Beijing city
PFASs
化合物含量(pg/m3) 2014年
10月
9日2014年
10月
13日2014年
10月
16日2014年
10月
18日2014年
10月
21日2014年
10月
23日2014年
10月
31日PFOS 9.7 1.3 0.9 0.8 3.7 10.8 3.6 FOSA 0.1 0.1 0.0 0.0 0.1 0.2 0.1 N-EtFOSA 0.5 0.5 0.6 0.6 0.3 0.5 0.5 PFHxA 1.6 0.4 0.5 0.6 0.6 1.4 0.7 PFHpA 2.6 0.2 0.2 0.5 0.7 2.3 0.6 PFOA 22.5 3.7 3.7 3.9 8.9 32.0 11.7 PFNA 3.0 1.4 1.8 2.3 1.0 2.7 1.2 PFDA 5.6 0.6 0.7 0.9 1.0 4.9 1.0 PFUnDA 1.6 0.6 0.7 0.6 0.8 1.6 0.8 PFDoDA 2.3 0.3 0.5 0.7 0.5 2.2 0.4 PFTrDA 1.1 0.3 0.3 0.7 0.6 0.9 0.4 PFTeDA 1.4 0.4 0.7 0.7 0.5 1.6 0.3 PFHxDA 1.0 0.2 0.4 0.4 0.8 1.1 0.5 PFOcDA 0.9 0.2 0.1 0.7 0.2 0.7 0.1 ∑PFASs 54.0 10.1 11.0 13.4 19.7 62.9 21.9 表 4 不同粒径下PFASs的q值
Table 4. q values of PFASs in different particle sizes
PFASs
化合物q值 >10 μm 2.5~
10 μm1~
2.5 μm0.25~
1 μm<0.25 μm PFOS 4.44 4.26 2.36 2.52 - FOSA - 9.64 7.18 6.45 9.40 N-EtFOSA 10.21 - 6.22 - - PFHxA 5.17 4.96 4.60 5.28 - PFHpA 4.86 4.03 4.49 5.03 4.85 PFOA 2.75 2.16 2.19 2.35 1.85 PFNA 5.18 4.66 4.80 4.82 - PFDA 5.07 4.73 4.16 4.19 3.40 PFUnDA 7.84 6.76 5.45 5.22 4.78 PFDoDA 7.46 - 5.37 4.74 4.37 PFTrDA 6.26 5.47 7.29 6.09 5.41 PFTeDA - - 6.38 5.40 5.26 PFHxDA 6.04 4.09 6.31 5.74 5.61 PFOcDA 7.79 - - 5.73 6.09 -
[1] Kissa E. Fluorinated Surfactants and Repellents (The Second Edition)[M]. New York: Marcel Dekker Inc., 2001.
[2] Cousins I T, Kong D, Vestergren R.Reconciling mea-surement and modelling studies of the sources and fate of perfluorinated carboxylates[J].Environmental Chemistry, 2011, 8(4):339-354.
[3] Shoeib M, Harner T, Vlahos P.Perfluorinated chemicals in the arctic atmosphere[J].Environmental Science & Technology, 2006, 40(24):7577-7583.
[4] Ding G, Xue H, Jing Z, et al.Occurrence and distribution of perfluoroalkyl substances(PFASs) in sediments of the Dalian Bay, China[J].Marine Pollution Bulletin, 2018, 127:285-288. doi: 10.1016/j.marpolbul.2017.12.020
[5] Wong F, Shoeib M, Katsoyiannis A, et al.Assessing temporal trends and source regions of per-and polyfluoroalkyl substances(PFASs) in air under the Arctic Monitoring and Assessment Programme (AMAP)[J].Atmospheric Environment, 2018, 172:65-73. doi: 10.1016/j.atmosenv.2017.10.028
[6] Guelfo J L, Adamson D T.Evaluation of a national data set for insights into sources, composition, and concentrations of per-and polyfluoroalkyl substances(PFASs) in U.S. drinking water[J].Environmental Pollution, 2018, 236:505-513. doi: 10.1016/j.envpol.2018.01.066
[7] Dalahmeh S, Tirgani S, Komakech A J, et al.Per-and polyfluoroalkyl substances (PFASs)in water, soil and plants in wetlands and agricultural areas in Kampala, Uganda[J].Science of the Total Environment, 2018, s631-632:660-667.
[8] 陈舒, 焦杏春, 盖楠, 等.中国东部农村地区土壤及水环境中全氟化合物的组成特征和来源初探[J].岩矿测试, 2015, 34(5):579-585. http://www.ykcs.ac.cn/article/doi/10.15898/j.cnki.11-2131/td.2015.05.014
Chen S, Jiao X X, Gai N, et al.Composition and source of perfluorinated compounds in soil and waters from the rural areas in Eastern China[J].Rock and Mineral Analysis, 2015, 34(5):579-585. http://www.ykcs.ac.cn/article/doi/10.15898/j.cnki.11-2131/td.2015.05.014
[9] 朴海涛, 陈舒, 焦杏春, 等.大运河丰水期水体中全氟化合物的分布[J].中国环境科学, 2016, 36(10):3040-3047. doi: 10.3969/j.issn.1000-6923.2016.10.029
Piao H T, Chen S, Jiao X X, et al.Geographical distribution of perfluorinated compounds in waters along the Grand Canal during wet season[J].China Environmental Science, 2016, 36(10):3040-3047. doi: 10.3969/j.issn.1000-6923.2016.10.029
[10] Odabasi M, Cetin B, Bayram A.Persistent organic po-llutants (POPs) on fine and coarse atmospheric particles measured at two (Urban and Industrial) sites[J].Aerosol & Air Quality Research, 2015, 15(5):1894-1905.
[11] Zhang X.Particle size distributions of PCDD/Fs and PBDD/Fs in ambient air in a suburban area in Beijing, China[J].Aerosol & Air Quality Research, 2015, 15(5):1933-1943.
[12] Dreyer A, Ebinghaus R.Polyfluorinated compounds in ambient air from ship-and land-based measurements inNorthern Germany[J].Atmospheric Environment, 2009, 43(8):1527-1535. doi: 10.1016/j.atmosenv.2008.11.047
[13] Genualdi S, Lee S C, Shoeib M, et al.Global pilot study of legacy and emerging persistent organic pollutants using sorbent-impregnated polyurethane foam disk passive air samplers[J].Environmental Science & Technology, 2010, 44(14):5534-5539.
[14] Kim S K, Shoeib M, Kim K S, et al.Indoor and outdoor poly-and perfluoroalkyl substances(PFASs) in Korea determined by passive air sampler[J].Environmental Pollution, 2012, 162(5):144-150.
[15] Barton C A, Butler L E, Zarzecki C J, et al.Characterizing perfluorooctanoate in ambient air near the fence line of a manufacturing facility:Comparing modeled and monitored values[J].Journal of the Air and Waste Management Association, 2006, 56(1):48-55. doi: 10.1080/10473289.2006.10464429
[16] Harada K, Nakanishi S, Sasaki K, et al.Particle size distribution and respiratory deposition estimates of airborne perfluorooctanoate and perfluorooctanesulfonate in Kyoto area, Japan[J].Bulletin of Environmental Contamination & Toxicology, 2006, 76(2):306-310.
[17] Dreyer A, Kirchgeorg T, Weinberg I, et al.Particle-size distribution of airborne poly-and perfluorinated alkyl substances[J].Chemosphere, 2015, 129(45):142-149.
[18] Ge H, Yamazaki E, Yamashita N, et al.Size specific distribution analysis of perfluoroalkyl substances in atmospheric particulate matter-development of a sampling method and their concentration in meeting room/ambient atmosphere[J].Aerosol & Air Quality Research, 2017, 17:553-562.
[19] Taniyasu S, Kannan K, Yeung L W, et al.Analysis of trifluoroacetic acid and other short-chain perfluorinated acids (C2-C4) in precipitation by liquid chromatography-tandem mass spectrometry:Comparison to patterns of long-chain perfluorinated acids (C5-C18)[J].Analytica Chimica Acta, 2008, 619(2):221-230. doi: 10.1016/j.aca.2008.04.064
[20] Baolin L, Hong Z, Dan Y, et al.Perfluorinated com-pounds (PFCs) in the atmosphere of Shenzhen, China:Spatial distribution, sources and health risk assessment[J].Chemosphere, 2015, 138:511-518. doi: 10.1016/j.chemosphere.2015.07.012
[21] Yao Y, Sun H, Gan Z, et al.A nationwide distribution of per-and polyfluoroalkyl substances (PFASs) in outdoor dust in mainland China from eastern to western areas[J].Environmental Science & Technology, 2016, 50(7):3676-3685.
[22] Guo M, Yan L, Xu T, et al.Particle size distribution and respiratory deposition estimates of airborne perfluoroalkyl acids during the haze period in the megacity of Shanghai[J].Environmental Pollution, 2018, 234:9-19. doi: 10.1016/j.envpol.2017.10.128
[23] 祝玉杰, 陈来国, 高博, 等.大气中全氟与多氟有机化合物研究进展[J].环境化学, 2015, 34(8):1396-1407. http://d.old.wanfangdata.com.cn/Periodical/hjhx201508002
Zhu Y J, Chen L G, Gao B, et al.Research advance on per-and poly-fluorinated organic compounds in the atmosphere[J].Environmental Chemistry, 2015, 34(8):1396-1407. http://d.old.wanfangdata.com.cn/Periodical/hjhx201508002
[24] Vestergren R, Cousins I T, Trudel D, et al.Estimating the contribution of precursor compounds in consumer exposure to PFOS and PFOA[J].Chemosphere, 2008, 73(10):1617-1624. doi: 10.1016/j.chemosphere.2008.08.011
[25] Schultz M M, Barofsky D F, Field J A.Fluorinated alkyl surfactants[J].Environmental Engineering Science, 2003, 20(5):487-501. doi: 10.1089/109287503768335959
[26] Taniyasu S, Yamashita N, Moon H B, et al.Does wet precipitation represent local and regional atmospheric transportation by perfluorinated alkyl substances?[J].Environment International, 2013, 55(4):25-32.
[27] Burns D C, Ellis D A, Li H, et al.Experimental pKa determination for perfluorooctanoic acid (PFOA) and the potential impact of pKa concentration dependence on laboratory-measured partitioning phenomena and environmental modeling[J].Environmental Science & Technology, 2008, 42(24):9283-9288.
[28] Cheng J, Psillakis E, Hoffmann M R, et al.Acid dissociation versus molecular association of perfluoroalkyl oxoacids:Environmental implications[J].Journal of Physical Chemistry A, 2009, 113(29):8152-8156. doi: 10.1021/jp9051352
[29] Goss K U, Arp H P.Comment on "Experimental pKa determination for perfluorooctanoic acid (PFOA) and the potential impact of pKa concentration dependence on laboratory-measured partitioning phenomena and envrionmental modeling"[J].Environmental Science & Technology, 2009, 43(13):5152-5154.
[30] Kutsuna S, Hori H, Sonoda T, et al.Preferential solvation of perfluorooctanoic acid (PFOA) by methanol in methanol-water mixtures:A potential overestimation of the dissociation constant of PFOA using a Yasuda-Shedlovsky plot[J].Atmospheric Environment, 2012, 49(49):411-414.
[31] Vierke L, Berger U, Cousins I T.Estimation of the acid dissociation constant of perfluoroalkyl carboxylic acids through an experimental investigation of their water-to-air transport[J].Environmental Science & Technology, 2013, 47(19):11032-11039.
[32] Arp H P, Schwarzenbach R P, Goss K U.Ambient gas/particle partitioning.1.Sorption mechanisms ofapolar, polar, and ionizable organic compounds[J].Environmental Science & Technology, 2008, 42(15):5541-5547.
[33] Yuan Q, Yang L, Dong C, et al.Particle physical characterisation in the Yellow River Delta of Eastern China:Number size distribution and new particle formation[J].Air Quality Atmosphere & Health, 2015, 8(5):441-452.
[34] Guo H, Wang D W, Cheung K, et al.Observation of aerosol size distribution and new particle formation at a mountain site in subtropical Hong Kong[J].Atmospheric Chemistry & Physics Discussions, 2012, 12(20):9923-9939. http://d.old.wanfangdata.com.cn/OAPaper/oai_doaj-articles_21918f3f99b44c27433af81df01a9853
[35] Cheng Y F, Su H, Rose D, et al.Size-resolved measurement of the mixing state of soot in the megacity Beijing, China:Diurnal cycle, aging and parameterization[J].Atmospheric Chemistry & Physics, 2013, 12(10):4477-4491. http://d.old.wanfangdata.com.cn/OAPaper/oai_doaj-articles_0cf5148cdfdddf1a427a1cb867f20ea1
-



 下载:
下载: