Dissolution Simulation Experiment of Buried Carbonate Reservoir: A Casefrom the Ordovician Carbonate Rock ofthe Western Ordos Basin
-
摘要:
通过模拟在埋藏条件下不同流体(0.2%的乙酸和分压2 MPa的CO2)与碳酸盐岩的溶蚀环境,对鄂尔多斯盆地奥陶系碳酸盐岩的溶蚀特性和微观特征进行研究。结果表明:在不同流体中白云岩溶蚀速率增加的幅度均高于石灰岩,在深埋藏环境下,二者的增加量差距更大,白云岩比石灰岩更易溶蚀形成溶蚀孔洞;乙酸中石灰岩和白云岩的溶蚀速率随温度压力的升高而升高,CO2中岩石的溶蚀速率先增后降;相同温度压力下,乙酸中岩石的溶蚀速率高于CO2中;岩石在CO2和乙酸溶液溶蚀后微观特征具有相似性,泥晶灰岩整体溶蚀降低,形成溶蚀坑;颗粒灰岩在原始的孔隙较发育的颗粒内部形成溶蚀坑;白云岩溶蚀后常形成晶间溶孔;白云质灰岩溶蚀后方解石整体溶蚀降低;硬石膏白云岩常形成膏模孔。实验证明埋藏条件下流体介质的差异对岩石溶蚀孔洞的发育具有重要影响。白云岩溶蚀能力大于石灰岩,溶蚀后孔隙发育,是马家沟组下组合值得关注的储集空间。
Abstract:The corrosion and microscopic characteristics of the Ordovician carbonate rocks in the Ordos Basin were studied by simulation of the carbonate dissolution in different fluids (0.2% acetic acid and CO2 under partial pressure of 2 MPa respectively) under buried conditions. It is found that the dissolution rates of dolomite in above fluids are obviously higher than those of limestone. In a deeply buried environment, the difference in dissolution rates of the two increases with depth. Dolomite is more likely to dissolve than limestone to form dissolved pores; The dissolution rates for both limestone and dolomite in acetic acid increase with temperature and pressure, while the dissolution rate of the two rocks increases first and then decreases afterwards in CO2. Under the same temperature and pressure, the dissolution rate of either dolomite or limestone is higher in acetic acid rather than in CO2; The microscopic characteristics of the rock after the dissolution in either CO2 or acetic acid are similar. For micritic limestone, dissolution is usually weak and easy to form corrosion pits; In granular limestone, however, corrosion pits are always found in the inner part of the grains; Intergranular pores are often formed by dissolution of dolomite; After dissolution of dolomite, the dissolution of calcite will slow down in general; Dissolved anhydrite mold pores are often observed in anhydritic limestone. The experiment results confirm that the fluid medium under burial conditions play important roles on the development of rock dissolution pores. The dissolution ability of dolomite is greater than that of limestone. The reservoir space developed by dissolution is worthy for attention in oil and gas exploration in the Majiagou Formation of the Ordos Basin.
-

-
表 1 溶蚀实验样品岩性及X衍射分析结果
Table 1. Lithology and X-ray diffraction analysis results of the samples for dissolution experiment
样号 井号 层位 深度/m 岩性 矿物种类和含量/% 石英 钾长石 斜长石 方解石 白云石 黄铁矿 赤铁矿 硬石膏 菱铁矿 黏土矿物 R-1 YT1 克里摩里组(马六段) 4 054.9 含云砂屑灰岩 1.77 / / 86.98 11.25 / / / / / R-2 ZT1 桌子山组(马五段、马四段) 4 353.2 白云质灰岩 1.88 / / 54.65 43.47 / / / / / R-3 ZT1 马五段 4 237.1 泥晶灰岩 7.86 / / 92.14 / / / / / / R-4 ZHT1 马四段 3 276.5 含生屑泥晶粉-砂屑灰岩 1.12 / / 95.28 3.59 / / / / / R-5 JT1 马五段 3 651.3 粉晶白云岩 / / / / 100.00 / / / / / R-6 DT1 马三段 4 416.55 硬石膏质泥晶白云岩 2.00 / / / 71.95 / / 26.05 / / 表 2 碳酸盐岩在2 mol/L乙酸溶液中的溶蚀实验数据记录
Table 2. Experimental Records of dissolution of carbonate rocks in acetic acid solution
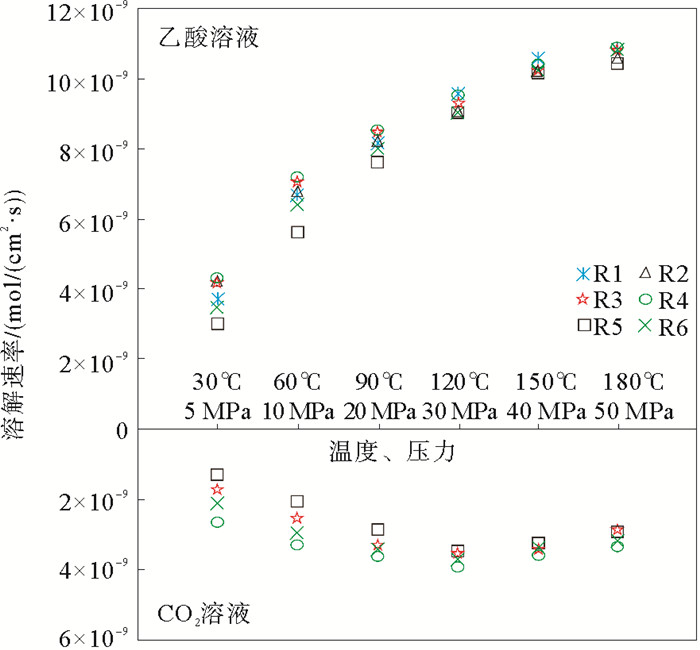
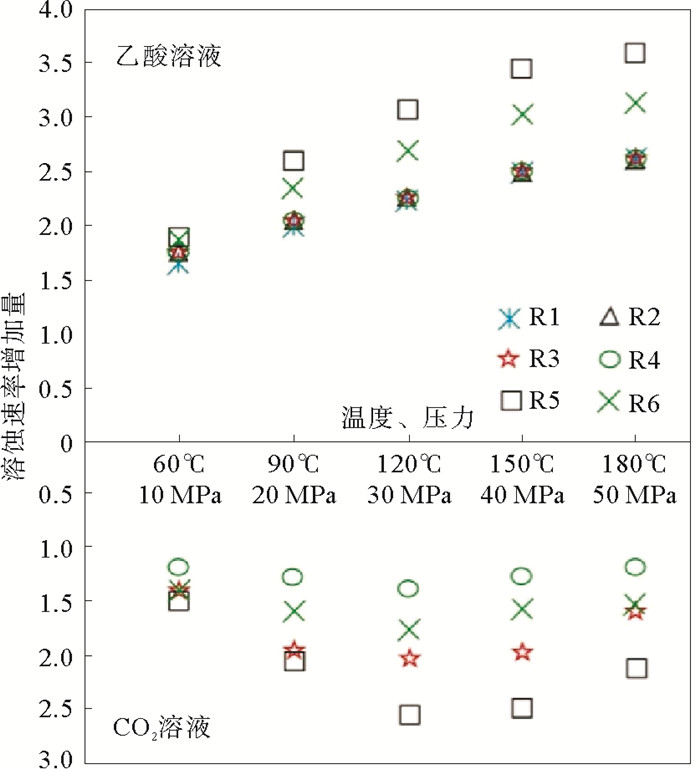
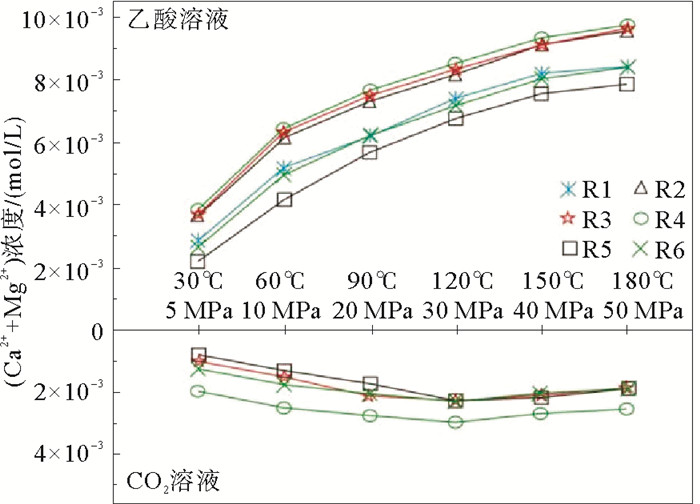
岩性 温度/℃ 压力/MPa Ca2+/(mol/L) Mg2+/(mol/L) (Ca2++Mg2+)/(mol/L) 表面积/cm2 溶解速度/(mol/(cm2·s)) 白云质灰岩 30 5 0.002 14 6.79×10-4 2.81×10-3 25.803 3.64×10-9 60 10 0.003 58 1.56×10-3 5.14×10-3 25.803 6.64×10-9 90 20 0.003 95 2.27×10-3 6.22×10-3 25.803 8.03×10-9 120 30 0.004 53 2.84×10-3 7.37×10-3 25.803 9.52×10-9 150 40 0.004 63 3.52×10-3 8.15×10-3 25.803 1.05×10-8 180 50 0.004 73 3.65×10-3 8.37×10-3 25.803 1.08×10-8 粉晶云岩 30 5 0.001 36 8.46×10-4 2.20×10-3 25.803 2.94×10-9 60 10 0.002 50 1.68×10-3 4.18×10-3 25.803 5.57×10-9 90 20 0.003 28 2.43×10-3 5.70×10-3 25.803 7.61×10-9 120 30 0.003 75 3.00×10-3 6.75×10-3 25.803 9.00×10-9 150 40 0.004 05 3.55×10-3 7.60×10-3 25.803 1.01×10-8 180 50 0.004 18 3.68×10-3 7.85×10-3 25.803 1.05×10-8 纹层状硬石膏质泥晶云岩 30 5 0.001 99 6.79×10-4 2.67×10-3 25.803 3.45×10-9 60 10 0.003 43 1.56×10-3 4.99×10-3 25.803 6.44×10-9 90 20 0.003 95 2.27×10-3 6.22×10-3 25.803 8.03×10-9 120 30 0.004 33 2.84×10-3 7.17×10-3 25.803 9.26×10-9 150 40 0.004 53 3.52×10-3 8.05×10-3 25.803 1.04×10-8 180 50 0.004 75 3.65×10-3 8.40×10-3 25.803 1.08×10-8 含云砂屑灰岩 30 5 0.003 63 3.17×10-5 3.66×10-3 29.964 4.07×10-9 60 10 0.006 00 6.46×10-5 6.06×10-3 29.964 6.75×10-9 90 20 0.007 20 8.75×10-5 7.29×10-3 29.964 8.11×10-9 120 30 0.008 00 1.18×10-4 8.12×10-3 29.964 9.03×10-9 150 40 0.008 93 1.49×10-4 9.07×10-3 29.964 1.01×10-8 180 50 0.009 33 1.60×10-4 9.48×10-3 29.964 1.06×10-8 砂屑灰岩 30 5 0.003 78 3.67×10-5 3.81×10-3 29.964 4.24×10-9 60 10 0.006 33 6.71×10-5 6.39×10-3 29.964 7.11×10-9 90 20 0.007 58 9.21×10-5 7.67×10-3 29.964 8.53×10-9 120 30 0.008 43 1.28×10-4 8.55×10-3 29.964 9.52×10-9 150 40 0.009 15 1.58×10-4 9.31×10-3 29.964 1.04×10-8 180 50 0.009 55 1.60×10-4 9.71×10-3 29.964 1.08×10-8 泥晶灰岩 30 5 0.003 68 3.29×10-5 3.71×10-3 29.964 4.12×10-9 60 10 0.006 38 6.79×10-5 6.44×10-3 29.964 7.17×10-9 90 20 0.007 48 9.13×10-5 7.57×10-3 29.964 8.42×10-9 120 30 0.008 25 1.27×10-4 8.38×10-3 29.964 9.32×10-9 150 40 0.009 00 1.54×10-4 9.15×10-3 29.964 1.02×10-8 180 50 0.009 48 1.60×10-4 9.64×10-3 29.964 1.07×10-8 表 3 碳酸盐岩在CO2(PCO2=2 MPa)中的溶蚀实验数据记录
Table 3. Records of dissolution experiment for carbonate rocks in CO2
岩性 温度/℃ 压力/MPa Ca2+/(mol/L) Mg2+/(mol/L) (Ca2++Mg2+)/(mol/L) 表面积/cm2 溶解速度/(mol/(cm2·s)) 含生屑泥粉晶-砂屑灰岩 30 5 0.002 01 2.92×10-5 2.04×10-3 25.907 2.63×10-9 60 10 0.002 55 3.21×10-5 2.58×10-3 25.907 3.32×10-9 90 20 0.002 75 3.46×10-5 2.84×10-3 25.907 3.58×10-9 120 30 0.002 98 3.79×10-5 2.84×10-3 25.907 3.88×10-9 150 40 0.002 800 2.75×10-5 3.07×10-3 25.907 3.64×10-9 180 50 0.002 55 3.38×10-5 2.58×10-3 25.907 3.32×10-9 泥晶灰岩 30 5 0.000 89 1.40×10-4 1.03×10-3 21.72 1.58×10-9 60 10 0.001 52 1.51×10-4 1.67×10-3 21.72 2.56×10-9 90 20 0.002 00 1.67×10-4 2.17×10-3 21.72 3.33×10-9 120 30 0.002 13 1.77×10-4 2.31×10-3 21.72 3.54×10-9 150 40 0.002 03 1.60×10-4 2.19×10-3 21.72 3.36×10-9 180 50 0.001 75 1.51×10-4 1.90×10-3 21.72 2.92×10-9 粉晶云岩 30 5 0.000 52 2.45×10-4 7.68×10-4 21.57 1.19×10-9 60 10 0.000 91 4.29×10-4 1.34×10-3 21.57 2.07×10-9 90 20 0.001 15 7.17×10-4 1.86×10-3 21.57 2.88×10-9 120 30 0.001 46 7.96×10-4 2.26×10-3 21.57 3.49×10-9 150 40 0.001 40 7.13×10-4 2.11×10-3 21.57 3.26×10-9 180 50 0.001 26 6.33×10-4 1.90×10-3 21.57 2.93×10-9 纹层状硬石膏质泥晶云岩 30 5 0.001 07 1.63×10-4 1.23×10-3 20.129 2.04×10-9 60 10 0.001 48 3.15×10-4 1.79×10-3 20.129 2.97×10-9 90 20 0.001 56 4.96×10-4 2.06×10-3 20.129 3.40×10-9 120 30 0.001 59 6.46×10-4 2.23×10-3 20.129 3.69×10-9 150 40 0.001 49 5.38×10-4 2.03×10-3 20.129 3.35×10-9 180 50 0.001 49 4.50×10-4 1.94×10-3 20.129 3.21×10-9 -
[1] 赵文智, 沈安江, 郑剑锋, 等.塔里木、四川及鄂尔多斯盆地白云岩储层孔隙成因探讨及对储层预测的指导意义[J].中国科学(地球科学), 2014, 44(9):1925-1939. http://d.old.wanfangdata.com.cn/Conference/8176860
[2] 覃建雄, 曽允孚.鄂尔多斯盆地东部下奥陶统碳酸盐岩成岩后生变化及孔隙演化[J].石油勘探与开发, 1993, 20(30):91-101. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK000002774576
[3] 覃建雄.鄂尔多斯盆地东部下奥陶统碳酸盐岩埋藏成岩事件研究[J].中国海上油气(地质), 1994, 8(1):45-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199400608676
[4] Weyl P K. The solution kinetics of calcite, The Journal of Geology, 1958, 66(2): 163-176. doi: 10.1086/626492
[5] Weyl P K. Porosity through dolomitization: Conservation-of-mass requirements, Journal of Sedimentary Research, 1960, 30(1): 85-90. http://d.old.wanfangdata.com.cn/Periodical/gwql201405013
[6] Akin G W, Lagerwerff J V. Calcium carbonate equilibria in solutions open to the air.Ⅱ. Enhanced solubility of CaCO3 in the presence of Mg2+ and SO-4 2[J]. Geochimica et Cosmochimica Acta, 1965, 29(4):353-360. doi: 10.1016/0016-7037(65)90026-8
[7] Plummer L N, Wigley T M L, Parkhurst D L. The kinetics of calcite dissolution in CO2-water systems at 5 to 60 ℃ and 0.0 to 1.0 atm CO2[J]. American Journal Science, 1978, 278(2): 179-216. doi: 10.2475/ajs.278.2.179
[8] Plummer L N, Parkhurst D L, Wigley T M L. Critical review of the kinetics of calcite dissolution and precipitation[C]//Jenne E A ed. Chemical Modeling in Aqueous Systems. Washington: American Chemical Society, 1979, 93: 537-573.
https://pubs.acs.org/doi/pdf/10.1021/bk-1979-0093.ch025 [9] Sjberg E L, Rickard D T. The influence of experimental design on the rate of calcite dissolution[J]. Geochimica et Cosmochimica Acta, 1983, 47(12): 2281-2285. doi: 10.1016/0016-7037(83)90051-0
[10] Sjberg E L, Rickard D T. Temperature dependence of calcite dissolution kinetics between 1 and 62℃ at pH 2.7 to 8.4 in aqueous solutions[J]. Geochimica et Cosmochimica Acta, 1984, 48(3): 485-493. doi: 10.1016/0016-7037(84)90276-X
[11] Chou L, Garrels R M, Wollast R. Comparative study of the kinetics and mechanisms of dissolution of carbonate minerals [J]. Chemical Geology, 1989, 78(3/4): 269-282. http://d.old.wanfangdata.com.cn/NSTLQK/10.1016-0009-2541(89)90063-6/
[12] 肖林萍, 黄思静.碳酸盐岩溶蚀实验热力学模型及工程地质意义[J].西南交通大学学报, 2002, 37(3):251-253. http://d.old.wanfangdata.com.cn/Periodical/xnjtdxxb200203006
[13] 肖林萍, 黄思静.方解石和白云石溶蚀实验热力学模型及地质意义[J].矿物岩石, 2003, 23(1):113-116. doi: 10.3969/j.issn.1001-6872.2003.01.024
[14] Pokrovsky O S, Golubev S V, Schott J. Calcite, dolomite and magnesite dissolution kinetics in aqueous solutions at acid to circumneutral pH, 25 to 150℃ and 1 to 55 atm PCO2: New constraints on CO2 Sequeatration in sedimentary basins[J]. Chemical Geology, 2009, 265(1/2): 20-32. https://www.sciencedirect.com/science/article/abs/pii/S0009254109000308
[15] 黄康俊, 王炜, 鲍征宇, 等.埋藏有机酸性流体对四川盆地东北部飞仙关组储层的溶蚀改造作用:溶解动力学实验研究[J].地球化学, 2011, 40(3):289-300. http://d.old.wanfangdata.com.cn/Periodical/dqhx201103008
[16] 杨俊杰, 黄思静, 张文正, 等.表生和埋藏成岩作用的温压条件下不同组成碳酸盐岩溶蚀成岩过程的实验模拟[J].沉积学报, 1995, 13(4):49-54. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199500000120
[17] 杨俊杰, 张文正, 黄思静, 等.埋藏成岩作用的温压条件下白云岩溶解过程的实验模拟研究[J].沉积学报, 1995, 13(3):83-88. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199500005866
[18] 黄思静, 杨俊杰, 张文正, 等.石膏对白云岩溶解影响的实验模拟研究[J].沉积学报, 1996, 14(1):103-109. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=QK199600006059
[19] Huang S J, Xiao L P, Yang J J, et al. Experimental Simulation of Dolomite Dissolution Under Burial Diagenesis Conditions and Thermodynamic Interpretation [J]. Chinese Journal of Geochemistry, 2000, 19 (1):58-64. doi: 10.1007/BF03166652
[20] 范明, 蒋小琼, 刘伟, 等.不同温度条件下CO2 水溶液对碳酸盐岩的溶蚀作用[J].沉积学报, 2007, 25(6):825-830. doi: 10.3969/j.issn.1000-0550.2007.06.002
[21] 佘敏, 朱吟, 沈安江, 等.塔中北斜坡鹰山组碳酸盐岩溶蚀的模拟实验研究[J].中国岩溶, 2012, 31(3):234-239. doi: 10.3969/j.issn.1001-4810.2012.03.002
[22] 佘敏, 寿建峰, 贺训云, 等.碳酸盐岩溶蚀机制的实验探讨:表面溶蚀与内部溶蚀对比[J].海相油气地质, 2013, 18(3):55-61. doi: 10.3969/j.issn.1672-9854.2013.03.007
[23] 佘敏, 寿建峰, 沈安江, 等.从表生到深埋藏环境下有机酸对碳酸盐岩溶蚀的实验模拟[J].地球化学, 2014, 43(3):277-286. http://d.old.wanfangdata.com.cn/Periodical/dqhx201403009
[24] 沈安江, 佘敏, 胡安平, 等.海相碳酸盐岩埋藏溶孔规模与分布规律初探[J].天然气地球科学, 2015, 26(10):1823-1830. doi: 10.11764/j.issn.1672-1926.2015.10.1823
[25] 佘敏, 寿建峰, 沈安江, 等.碳酸盐岩溶蚀规律与孔隙演化实验研究[J].石油勘探与开发, 2016, 43(4):564-572. http://d.old.wanfangdata.com.cn/Periodical/syktykf201604008
[26] 寿建峰, 佘敏, 沈安江.深层条件下碳酸盐岩溶蚀改造效应的模拟实验研究[J].矿物岩石地球化学通报, 2016, 35(5):860-867, 806. doi: 10.3969/j.issn.1007-2802.2016.05.006
[27] 钱一雄, Conxita T, 邹森林, 等.碳酸盐岩表生岩溶与埋藏溶蚀比较——以塔北和塔中地区为例[J].海相油气地质, 2007, 12(2):1-7. doi: 10.3969/j.issn.1672-9854.2007.02.001
[28] 范明, 胡凯, 蒋小琼, 等.酸性流体对碳酸盐岩储层的改造作用[J].地球化学, 2009, 38(1):20-26. doi: 10.3321/j.issn:0379-1726.2009.01.002
[29] 谭飞, 张云峰, 王振宇, 等.鄂尔多斯盆地奥陶系不同组构碳酸盐岩埋藏溶蚀实验[J].沉积学报, 2017, 35(2):413-424. http://d.old.wanfangdata.com.cn/Periodical/cjxb201702018
[30] Cubilias P, Khler S, Prieto M, et al. How do mineral coatings affect dissolution rates? An experimental study of coupled CaCO3 dissolution-CdCO3 precipitation [J].Geochim Cosmochim Acta, 2005, 69(23): 5459-5476. doi: 10.1016/j.gca.2005.07.016
[31] Gong Q J, Deng J, Wang Q F, et al. Calcite dissolution in deionized water from 50℃ to 250℃ at 10 MPa: Rate equation and reaction ordor [J]. Acta geologica Sinica, 2008, 82(5):994-1001. http://en.cnki.com.cn/Article_en/CJFDTOTAL-DZXW200805009.htm
[32] 黄思静, 黄培培, 黄可可, 等.碳酸盐倒退溶解模式的化学热力学基础——与H2S有关的溶解介质及其与CO2的对比[J].沉积学报, 2010, 28(1):1-9. http://www.wanfangdata.com.cn/details/detail.do?_type=perio&id=cjxb201001001
[33] Davies G R, Smith J L B. Structurally controlled hydrothermal dolomite reservoir facies: An overview [J]. AAPG Bulletin, 2006, 90(11): 1641-1690. doi: 10.1306/05220605164
[34] 黄思静, 黄可可, 张雪花, 等.碳酸盐倒退溶解模式的化学热力学基础——与CO2有关的溶解介质[J].成都理工大学学报(自然科学版), 2009, 36(5):457-464. doi: 10.3969/j.issn.1671-9727.2009.05.001
[35] 于洲, 孙六一, 吴兴宇, 等.鄂尔多斯盆地靖西地区马家沟组中组合储层特征及主控因素[J].海相油气地质, 2012, 17(4):49-56. doi: 10.3969/j.issn.1672-9854.2012.04.008
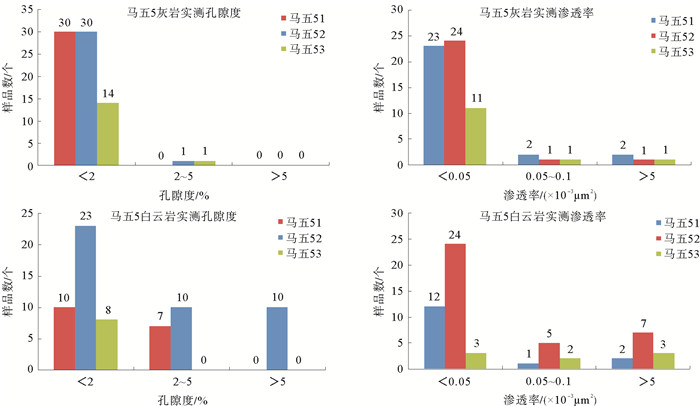
[36] 朱广社, 赵俊兴, 邵晓岩, 等.苏里格东部马五5储层特征及成藏控制因素[J].西南石油大学学报(自然科学版), 2014, 36(3):45-53.
[37] 时保宏, 刘亚楠, 武春英, 等.鄂尔多斯盆地古隆起东侧奥陶系马家沟组中组合成藏地质条件[J].石油与天然气地质, 2013, 34(5):610-618. http://d.old.wanfangdata.com.cn/Periodical/syytrqdz201305005
[38] 吴东旭, 吴兴宁, 曹荣荣, 等.鄂尔多斯盆地奥陶系古隆起东侧马家沟组中组合储层特征及成藏演化[J].海相油气地质, 2014, 19(4):38-44. doi: 10.3969/j.issn.1672-9854.2014.04.006
-



 下载:
下载: