Study on the Interface Adsorption Behavior of Diester Gemini Quaternary Ammonium Salt on the Fluorapatite and Quartz
-
摘要:
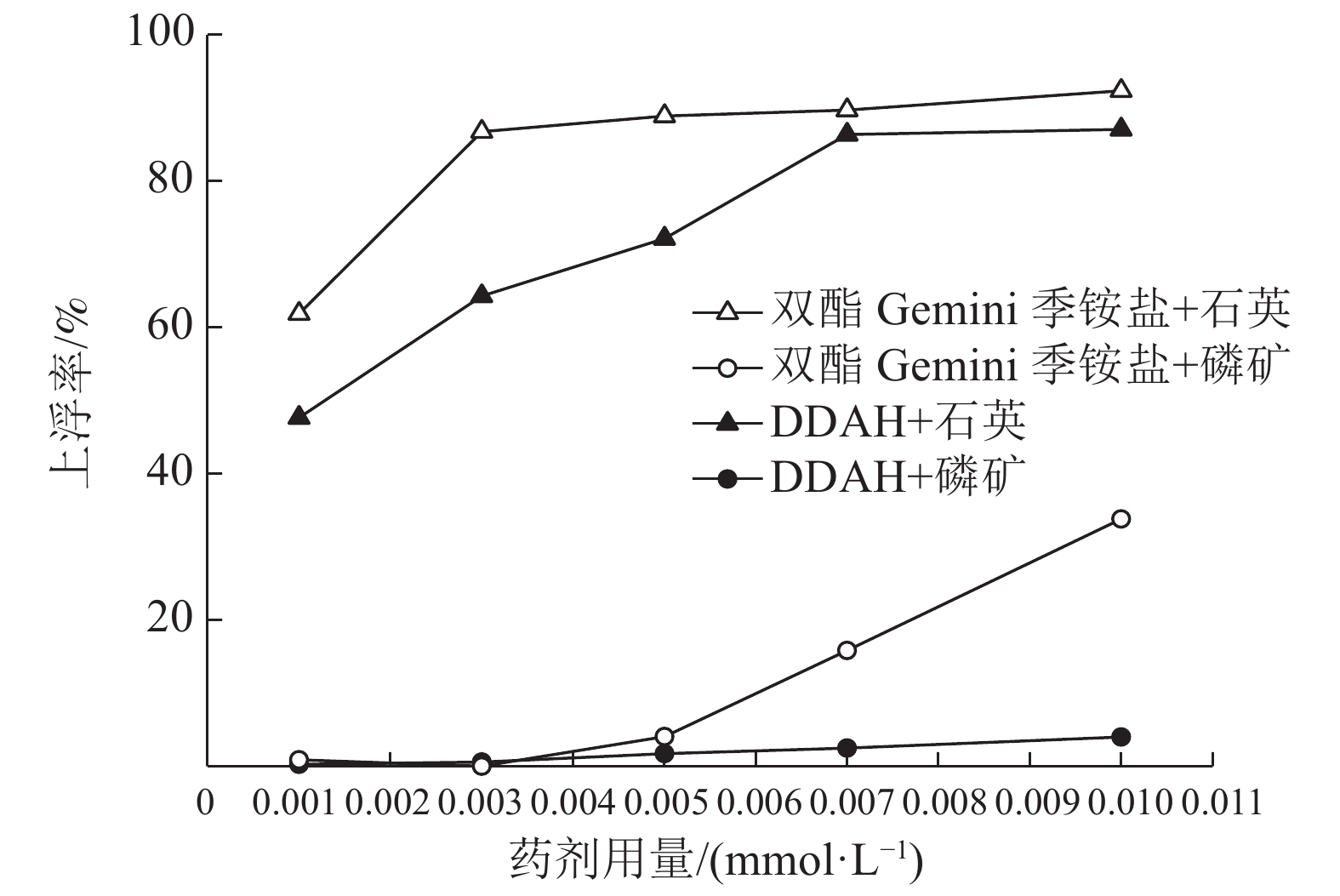
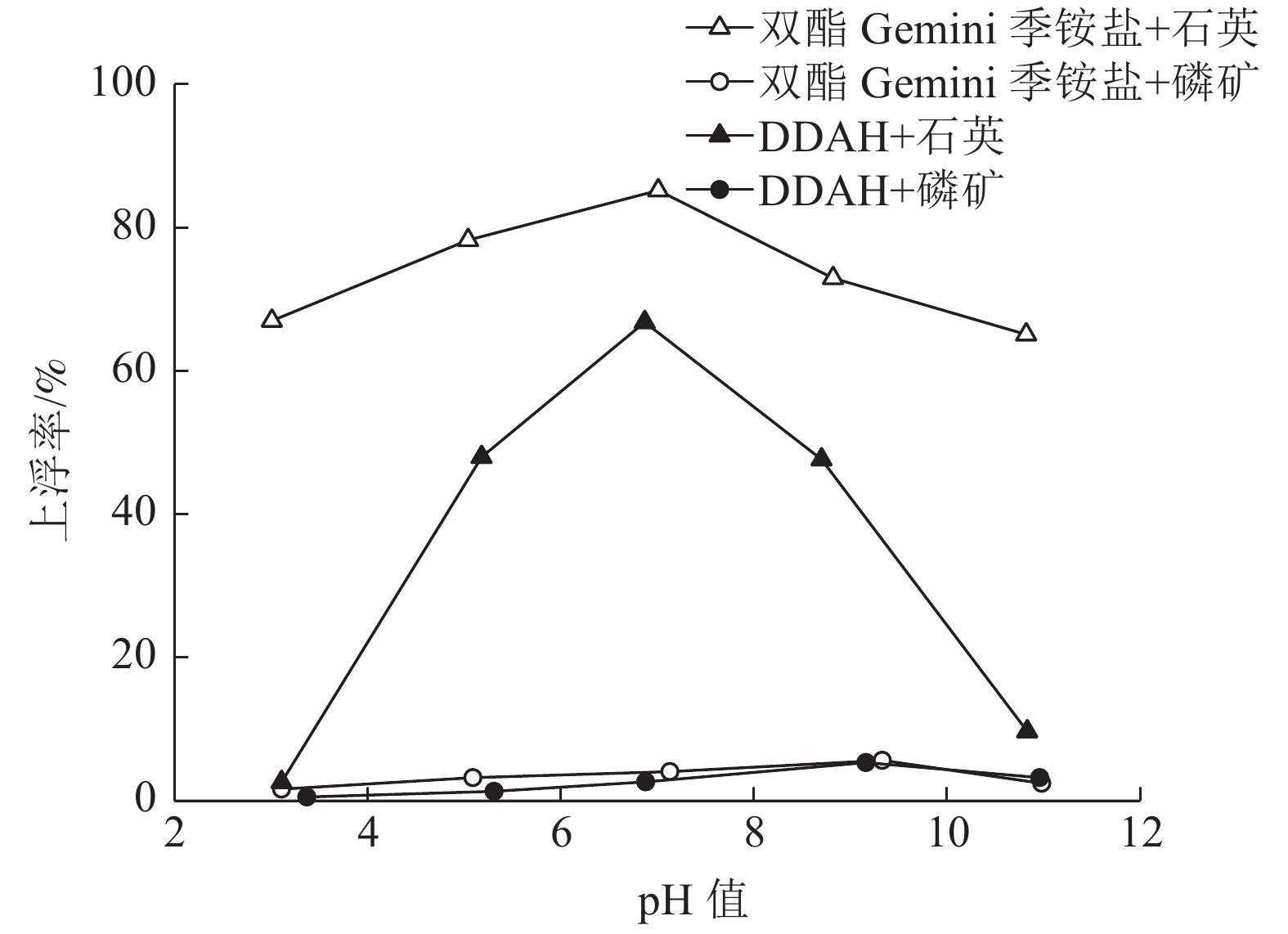
对自制双酯Gemini季铵盐进行表面张力测试,量子化学计算,以及单矿物浮选实验,结果表明自制双酯Gemini季铵盐与十二胺盐酸盐(DDAH)相比,具有更低的CMC,更强的阳离子性,更大的分子电负性,并且在低浓度5×10-6mol/L时,双酯Gemini季铵盐对石英单矿物的浮选上浮率可以达到88.89%,磷灰石上浮率仅为4.04%。通过接触角测试、分子动力学模拟,研究双酯Gemini季铵盐在石英、磷矿界面上的吸附过程,结果表明,相比磷灰石,双酯Gemini季铵盐更易于在石英表面吸附,可作为石英和氟磷灰石浮选分离的捕收剂。
-
关键词:
- 双酯Gemini季铵盐 /
- 石英 /
- 氟磷灰石 /
- 分子动力学模拟
Abstract:Surface tension tests, quantum chemical computation and flotation tests were carried on the self-synthesized Diester Gemini quaternary ammonium salt. The results indicated that Diester Gemini quaternary ammonium salt has lower CMC, stronger cationic, larger molecular electro negativity, and at the low concentration of 5×10-6 mol/L, resulting in quartz recovery at 88.89%, fluorapatite recovery at 4.04%. According to the results of contact angle tests and molecular dynamics simulations, Diester Gemini quaternary ammonium salt prefer to adsorb on quartz surface than fluorapatite. Thus, the Diester Gemini quaternary ammonium salt can be used as a cationic collector for quartz from fluorapatite.
-

-
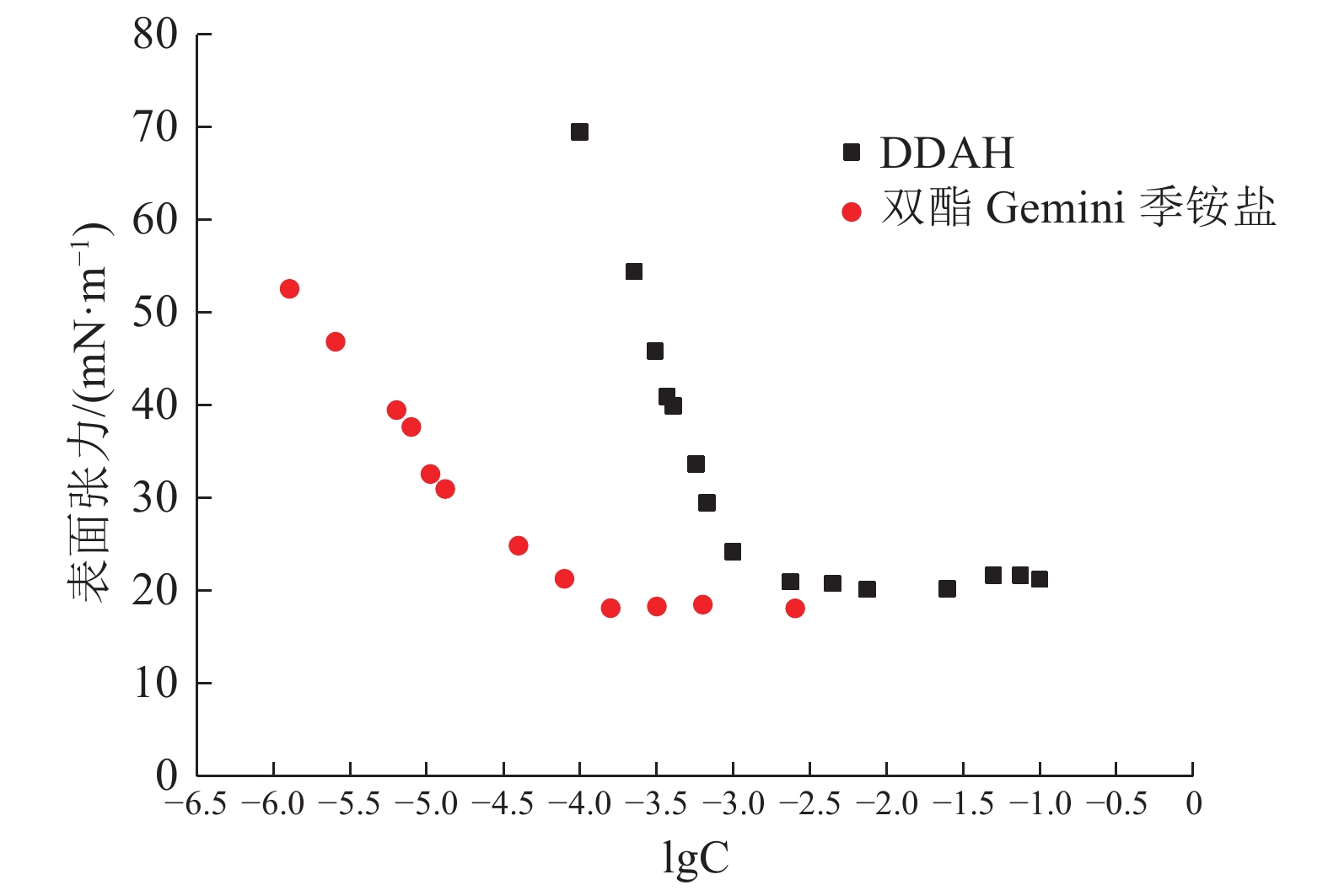
表 1 DDAH与双酯Gemini季铵盐表面性能计算结果
Table 1. Results of surface properties tests of DDAH and Diester Gemini quaternary ammonium salt
药剂 CMC/(mol·L-1) γcmc/(mN·m-1) Γmax/(mol·m-2) Am/m2 DDAH 1.096×10-3 20.31 5.563×10-3 0.299×10-21 双酯Gemini季铵盐 0.912×10-4 18.30 2.649×10-3 0.627×10-21 表 2 药剂与矿物的前线轨道能与原子Mulliken布局计算结果/a.u.
Table 2. Frontier orbital eigenvalues and some selected Mulliken charges of reagents and minerals
药剂、矿物 原子Mulliken电荷 基团电荷 LUMO HOMO |ΔE药剂-石英| |ΔE药剂-磷矿| 分子绝对电负性 DDAH N:-0.259 NH3+:0.597 -0.195 -0.318 0.108 0.103 0.257 双酯Gemini
季铵盐N:-0.442;-0.426
O:-0.471;-0.484CH2N+(CH3)2CH2:0.780;0.795
OH:-0.180;-0.197-0.201 -0.334 0.102 0.097 0.268 石英 -0.077 -0.303 磷矿 -0.080 -0.298 表 3 双酯Gemini季铵盐对石英(101)面、氟磷灰石(001)面的吸附作用能/(kcal·mol-1)
Table 3. Interaction energies on quartz (101) surface and fluorapatite (001) surface
矿物-药剂 E药剂与矿物 E药剂 E矿物 ΔE 石英-双酯Gemini季铵盐 11483.295 373.321 11162.002 -52.028 磷矿-双酯Gemini季铵盐 2989.222 351.211 2679.216 -41.205 石英-DDAH 1923.619 101.229 1838.613 -16.223 磷矿-DDAH 48.992 87.104 -24.003 -14.109 -
[1] 石天宇, 卯松, 李先海, 等. 调整剂对胶磷矿和石英矿浆表面张力的影响研究[J]. 化工矿物与加工, 2016(5):2-4+11. SHI T Y, MAO S, LI X H, et al. Study on the effect of adjusters on the surface tension of phosphate rock and quartz slurry[J]. Chemical Minerals and Processing, 2016(5):2-4+11. doi: 10.16283/j.cnki.hgkwyjg.2016.05.001
[2] 曾小波, 葛英勇. 胶磷矿阳离子反浮选泡沫行为调控研究[J]. 化工矿物与加工, 2008, 1: 1-3.
ZENG X B, GE Y Y. Study on the regulation of foam behavior in cationic reverse flotation of phosphate rock [J] Chemical Minerals and Processing, 2008, 1: 1-3.
[3] 张富青, 王巍, 潘志权, 等. 一种新型磷矿反浮选硅酸盐捕收剂机理研究[J]. 中国矿业, 2013, 22(8):108-111. ZHANG F Q, WANG W, PAN Z Q, et al. Study on the mechanism of a new type of silicate collector for reverse flotation of phosphate rock[J]. China Mining, 2013, 22(8):108-111. doi: 10.3969/j.issn.1004-4051.2013.08.027
[4] 吴中贤, 姜效军, 陶东平. 新型胶磷矿反浮选脱硅阳离子捕收剂试验研究[J]. 矿产综合利用, 2020(5):115-119. WU Z X, JIANG X J, TAO D P. Experimental study on a novel cationic collector for reverse flotation of collophane for silica removal[J]. Multipurpose Utilization of Mineral Resources, 2020(5):115-119. doi: 10.3969/j.issn.1000-6532.2020.05.017
[5] 叶军建. 微细粒磷灰石浮选的界面调控研究[D]. 贵阳: 贵州大学, 2019.
YE J J. Study on interface control of fine apatite flotation [D]. Guiyang: Guizhou University, 2019.
[6] 李冬莲, 卢寿慈, 谢恒星. 磷灰石常温浮选溶液化学的研究[J]. 矿冶工程, 1999(1):35-37. LI D L, LU S C, XIE H X. Study on the chemistry of apatite flotation solution at room temperature[J]. Mining and Metallurgical Engineering, 1999(1):35-37.
[7] 朱一民. 2019年浮选药剂的进展[J]. 矿产综合利用, 2020(5):1-17. ZHU Y M. Development of flotation reagent in 2019[J]. Multipurpose Utilization of Mineral Resources, 2020(5):1-17. doi: 10.3969/j.issn.1000-6532.2020.05.001
[8] Jacek Luczyński, Frackowiak R, Wloch A, et al. Gemini ester quat surfactants and their biological activity[J]. Cellular & Molecular Biology Letters, 2013, 18(1):89-101.
[9] 钟宣. 浮选药剂的结构与性能—浮选药剂性能的CMC计算法[J]. 有色金属(冶炼部分), 1977(6):25-28. ZHONG X. Structure and performance of flotation reagent - CMC calculation method of flotation reagent performance[J]. Nonferrous Metals (Extractive Metallurgy), 1977(6):25-28.
[10] 钟宣. 浮选药剂的结构与性能——浮选药剂分子几何大小与选择性[J]. 有色金属(冶炼部分), 1977(10):13-20. ZHONG X. Structure and performance of flotation reagents - molecular geometric size and selectivity of flotation reagents[J]. Nonferrous Metals(Extractive Metallurgy), 1977(10):13-20.
[11] 陈建华, 冯其明, 卢毅屏. 浮选药剂亲固基团的设计[J]. 有色金属工程, 1999(2):19-23. CHEN J H, FENG Q M, LU Y P. Design of solid group of flotation reagent[J]. Nonferrous Metal Engineering, 1999(2):19-23.
[12] 周国华, 孙伟, 薛玉兰, 等. 化学反应电子转移数判据在浮选捕收剂结构与性能关系的应用研究[J]. 有色金属工程, 2001, 53(1):19-22. ZHOU G H, SUN W, XUE Y L, et al. Study on the application of chemical reaction electron transfer number criterion in the relationship between structure and performance of flotation collectors[J]. Nonferrous Metal Engineering, 2001, 53(1):19-22. doi: 10.3969/j.issn.2095-1744.2001.01.005
[13] 王国芝, 徐刚, 徐盛明, 等. 浮选药剂结构与性能关系的研究进展[J]. 矿产保护与利用, 2012(1):53-58. WANG G Z, XU G, XU S M, et al. Research progress on the relationship between structure and performance of flotation reagents[J]. Conservation and Utilization of Mineral Resources, 2012(1):53-58. doi: 10.3969/j.issn.1001-0076.2012.01.014
[14] Liu W, Liu W, Zhao B, et al. Novel insights into the adsorption mechanism of the isopropanol amine collector on magnesite ore: A combined experimental and theoretical computational study[J]. Powder Technology, 2019, 343:366-374. doi: 10.1016/j.powtec.2018.11.063
[15] Li L, Hao H, Yuan Z, et al. Molecular dynamics simulation of siderite-hematite-quartz flotation with sodium oleate[J]. Applied Surface Science, 2017, 419(15):557-563.
[16] 南楠, 朱一民, 韩跃新, 等. 基于CASTEP模拟的氟磷灰石与十二烷基磷酸酯作用机理研究[J]. 金属矿山, 2020, 528(6):105-109. NAN N, ZHU Y M, HAN Y X, et al. Study on the interaction mechanism of fluoroapatite and dodecyl phosphate based on CASTEP simulation[J]. Metal Mines, 2020, 528(6):105-109. doi: 10.19614/j.cnki.jsks.202006015
[17] 刘凤霞. 氧化铅浮选黄药分子结构与性能研究[D]. 南宁: 广西大学, 2007.
LIU F X. Study on the molecular structure and properties of xanthate in lead oxide flotation [D]. Nanning: Guangxi University, 2007.
-



 下载:
下载: