Effect of High Temperature Roasting on Heavy Metal Adsorption of Bauxite Tailings
-
摘要:
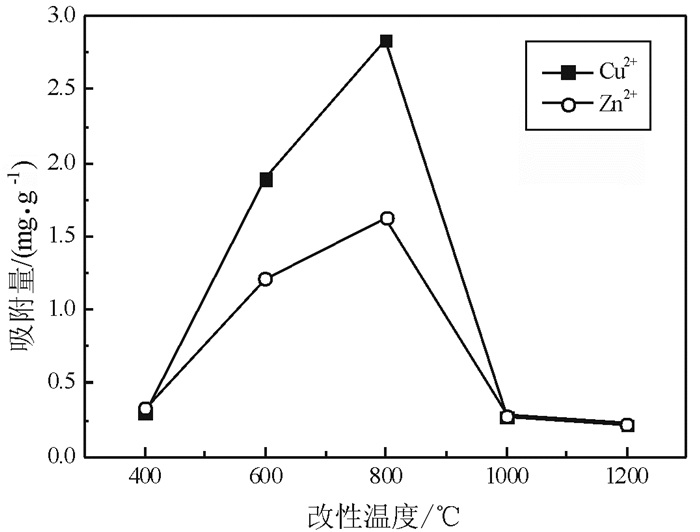
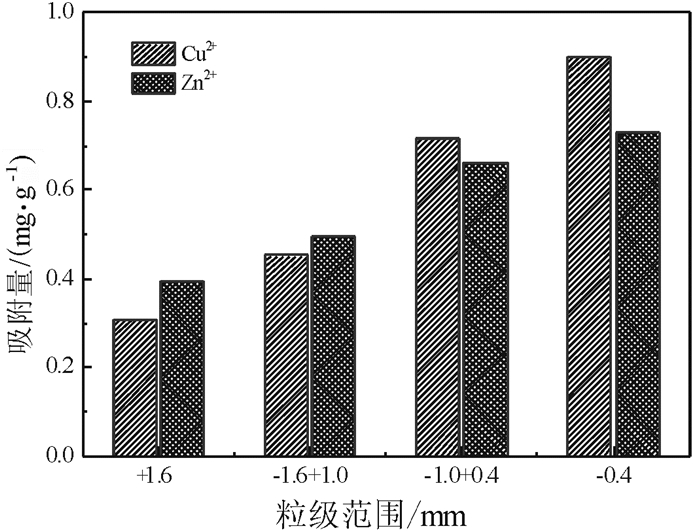
我国每年均消耗大量的铝土矿资源,由此产生了大量的铝土矿尾矿。与此同时,尾矿堆积而产生的环境污染问题越来越为人们所重视。铝土矿尾矿中多含有高岭石、伊利石、叶蜡石等层状硅酸盐矿物,具有良好的多孔性能,可作为废水处理剂使用。鉴于此,为实现铝土矿尾矿资源的综合利用,采用高温焙烧方法对铝土矿尾矿进行了改性研究,并考察了改性后的铝土矿尾矿对重金属离子Cu2+和Zn2+吸附性能。试验表明:在焙烧温度为800 ℃条件下连续焙烧2 h后,该尾矿具有最好的吸附效果:常温条件下,对Cu2+和Zn2+的饱和吸附量达到2.837 mg/g和1.630 mg/g。以上研究表明,高温焙烧可以促进铝土矿尾矿对重金属的吸附性能,有可能实现该尾矿的综合利用。
Abstract:Large amounts of bauxite resources are consumed annually in China, which produces many tailings. Furthermore, the accumulation of these tailings has caused series environmental problems and drawn more attention. Bauxite tailings contain kaolinite, illite, pyrophyllite and other layered silicate minerals, which have good porous properties for wastewater treatment. In order to solve the environmental problems of bauxite tailings and facilitate the comprehensive utilization of these resources, high temperature roasting modification was applied to enhance the adsorption performance of Cu2+ and Zn2+. The results indicated that the best adsorption results with saturated adsorption capacity of 2.837 mg/g and 1.630 mg/g for Cu2+ and Zn2+ could be achieved when the tailing was roasted at 800 ℃ for 2 h. Based on these results, it is found that heavy metal adsorption ability of bauxite tailings could be improved by high temperature modification, which make it possible to realize the comprehensive utilization of the tailings.
-
Key words:
- bauxite tailings /
- high temperature modification /
- adsorption /
- copper ion /
- zinc ion
-

-
表 1 -0.4 mm粒级铝土矿尾矿焙烧前后对重金属的吸附性能
Table 1. Heavy metal adsorption amounts on -0.4 mm fraction tailing before and after being roasted
饱和吸附量/(mg·g-1) 改性前 800 ℃改性后 铜离子 0.902 2.837 锌离子 0.732 1.630 -
[1] 姜涛, 邱冠周, 李光辉, 等.中低品位铝土矿选矿预脱硅的新进展[J].矿冶工程, 1996, 19(2):3-6. http://industry.wanfangdata.com.cn/yj/Detail/Patent?id=Patent_CN200510124012.6&type=Free
[2] 张汉平, 李福全.云南某堆积型低品位铝土矿浮选脱硅试验研究[J].云南冶金, 2015, 44(4):10-14. http://www.cqvip.com/QK/97765X/201502/664478685.html
[3] 周长春, 刘炯天, 李振, 等.中低品位铝土矿浮选柱短流程分选研究[J].中国矿业大学学报, 2010, 39(1):116-120. https://www.wenkuxiazai.com/doc/05a62276a98271fe910ef9ce.html
[4] 卢清华, 胡岳华.铝土矿选矿尾矿特性[J].矿物学报, 2012, 32(4):537-542. http://www.cqvip.com/QK/90627X/201406/50264015.html
[5] 鲁丰春, 王鹏.河南低品位铝土矿工艺矿物学分析[J].轻金属, 2011(s1):43-45. https://www.wenkuxiazai.com/doc/c9cc2a08f18583d048645910.html
[6] 王鹏, 王宝奎, 石建军, 等.低品位铝土矿选矿技术的优化[J].轻金属, 2011(9):8-10. http://www.cqvip.com/QK/91164X/201109/39512054.html
[7] 兰叶, 王毓华, 胡业民.铝土矿浮选尾矿基本特性与再利用研究[J].轻金属, 2006(10):9-12. doi: 10.3969/j.issn.1002-1752.2006.10.003
[8] 何宏平, 郭九皋, 朱建喜, 等.蒙脱石、高岭石、伊利石对重金属离子吸附容量的实验研究[J].岩石矿物学杂志, 2001, 20(4):573-578. https://www.wenkuxiazai.com/doc/a9d63cfd52ea551811a687be.html
[9] Atapour H. Geochemistry of potentially harmful elements in topsoils around Kerman city, southeastern Iran[J]. Environmental Earth Sciences, 2015, 74 (7): 5605-5624. doi: 10.1007/s12665-015-4576-3
[10] 罗琳, 刘永康, 何伯泉.一水硬铝石-高岭石型铝土矿焙烧脱硅热力学机理研究[J].有色金属, 1999, 51(1):25-30. http://industry.wanfangdata.com.cn/dl/Detail/Thesis?id=Thesis_Y290245
[11] 范晓慧, 李光辉, 姜涛, 等.铝土矿焙烧脱硅新工艺及机理研究[J].金属矿山, 2002(7):16-18. http://doi.wanfangdata.com.cn/10.3321/j.issn:1001-1250.2002.07.006
-



 下载:
下载: