Determination of Hexavalent Chromium in Soil by Inductively Coupled Plasma-Mass Spectrometry with Alkaline Digestion-Ion Exchange
-
摘要:
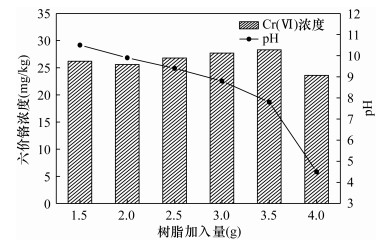
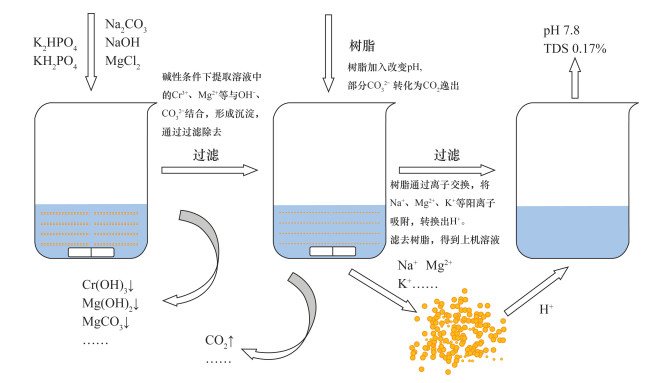
六价铬Cr(Ⅵ)是建设用地土壤基本监测指标之一,开展土壤六价铬监测在环境污染防治中具有重要意义。目前测定土壤Cr(Ⅵ)的标准方法为火焰原子吸收分光光度法(FAAS),其检出限为0.5mg/kg,难以满足低浓度土壤样品中Cr(Ⅵ)的分析。本文采用氢氧化钠-碳酸钠溶液提取土壤中的Cr(Ⅵ),建立了一种使用电感耦合等离子体质谱法(ICP-MS)结合阳离子交换测定土壤中低浓度Cr(Ⅵ)的方法。结果表明,将碱性提取液稀释10倍并加入3.5g阳离子交换树脂后,溶解性总固体(TDS)质量分数从2.4%降低为0.17%,基体干扰大幅度降低。同时,由于离子交换过程溶出氢离子,使得pH达到适宜的检测范围(7.5±0.5)。此外,优化了前处理条件,提取温度为90~95℃、搅拌速度300r/min、加热时间70min时,Cr(Ⅵ)提取效果最佳,相对误差(RE)仅-1.7%。本方法相对标准偏差(RSD)为3.1%~5.9%,平均相对误差介于-3.8%~-1.1%。使用F检验及t检验比较了ICP-MS法与FAAS法测试高、中、低三种浓度标准物质的结果,二者无显著性差异。由于本法采取稀释、离子交换、内标法等方式降低了基体干扰,结合ICP-MS自身灵敏度高、准确性好的特点,使得方法检出限(MDL)达到0.061mg/kg,显著低于FAAS法检出限(0.5mg/kg),可用于低浓度土壤Cr(Ⅵ)样品的测定。
-
关键词:
- 电感耦合等离子体质谱法 /
- 火焰原子吸收分光光度法 /
- 离子交换 /
- 氢氧化钠-碳酸钠溶液提取 /
- 六价铬
Abstract:BACKGROUND Hexavalent chromium Cr(Ⅵ) is one of the basic monitoring indicators of soil in construction land. It is of great significance to carry out soil hexavalent chromium monitoring in the prevention and control of environmental pollution. At present, the standard method of hexavalent chromium in soil is flame atomic absorption spectrometry (FAAS). The FAAS method has a high detection limit (0.5mg/kg), and serious matrix effect, and cannot meet the analysis of Cr(Ⅵ) in low-concentration soil samples.
OBJECTIVES To establish a convenient and high sensitivity method for determination of low-concentration hexavalent chromium in soil.
METHODS An ion-exchange-inductively coupled plasma-mass spectrometry (ICP-MS) method was developed to determine the content of hexavalent chromium in soil by extracting hexavalent chromium with alkali solution. The resin content, mixing speed, extraction temperature and extraction time were studied. The measurement results were compared with the flame atomic absorption spectrophotometry (FAAS) method.
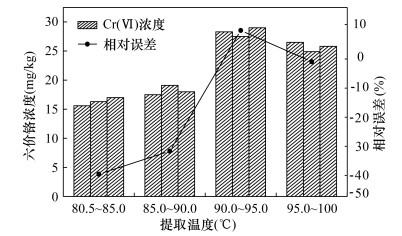
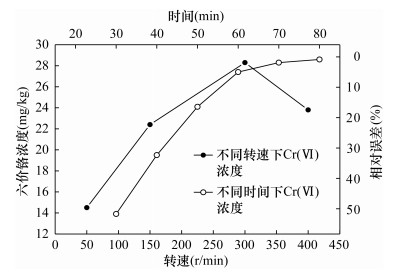
RESULTS The results showed that the total dissolved solids (TDS) mass fraction was reduced from 2.4% to 0.17% after the alkaline extraction solution was diluted 10 times and 3.5g of cation exchange resin was added, and the matrix interference was greatly reduced. At the same time, due to the dissolution of hydrogen ions in the ion exchange process, the pH reached a suitable detection range (7.5±0.5). The pretreatment conditions were optimized. When the extraction temperature was 90-95℃, the stirring speed was 300 rpm, and the heating time was 70 min, the extraction effect of Cr(Ⅵ) was the best, and the relative error was -1.7%. The relative standard deviation (RSD) was 3.1%-5.9%, and the average relative error was -3.8% to -1.1%. F test and t test were used to compare the test results of high, medium and low concentration standard substances by ICP-MS and FAAS, and there was no significant difference between the two methods. The method detection limit (MDL) was 0.061 mg/kg.
CONCLUSIONS Since this method adopts dilution, ion exchange, internal standard method, to reduce matrix interference, combined with the high sensitivity and good accuracy of ICP-MS, the method detection limit (MDL) is significantly lower than the detection limit of FAAS method (0.5mg/kg). This method can be used for the determination of low-concentration soil Cr(Ⅵ) samples.
-

-
表 1 采用ICP-MS和FAAS法测定土壤中Cr(Ⅵ)的检出限
Table 1. Detection limit of Cr(Ⅵ) in soil by ICP-MS and FAAS
分析方法 提取液平行测定的Cr(Ⅵ)浓度(mg/L) 标准偏差(mg/kg) 方法检出限MDL(mg/kg) 1 2 3 4 5 6 7 ICP-MS(本文方法) 0.1902 0.2004 0.1578 0.1822 0.1736 0.2148 0.1690 0.01949 0.061 FAAS 2.06 1.90 1.72 1.84 1.68 2.02 1.78 0.145 0.5 表 2 采用ICP-MS和FAAS法测定Cr(Ⅵ)的精密度及相对误差
Table 2. Precision and relative error of ICP-MS and FAAS determination of Cr(Ⅵ)
标准物质编号 Cr(Ⅵ)浓度标准值(mg/kg) 分析方法 土壤中Cr(Ⅵ)浓度平行测定值(mg/kg) 相对误差(%) RSD(%) 1 2 3 4 5 6 7 平均值 RMH-A048 28.8±2.7 ICP-MS 27.1 28.3 26.7 29 27.5 28.3 26.9 27.7 -3.8 3.1 FAAS 28.3 31.1 26.9 27.5 28.3 26.8 27.7 28.1 -2.4 5.2 RMU041 5.10±0.67 ICP-MS 5.11 4.93 5.02 4.67 4.52 4.81 5.30 4.91 -3.7 5.4 FAAS 4.39 5.73 4.52 4.39 4.64 5.13 4.45 4.75 -6.9 10.6 GBW(E)070251 0.92±0.09 ICP-MS 0.84 0.86 0.93 0.89 0.96 0.99 0.92 0.91 -1.1 5.9 FAAS 0.70 0.83 0.96 0.82 0.96 0.91 0.63 0.83 -9.8 15.3 -
[1] Chen J S, Wei F S, Zheng C J, et al. Background concentrations of elements in soils of China[J]. Water Air and Soil Pollution, 1991, 57/58(1): 699-712. doi: 10.1007/BF00282934
[2] 陈雅丽, 翁莉萍, 马杰, 等. 近十年中国土壤重金属污染源解析研究进展[J]. 农业环境科学学报, 2019, 38(10): 2219-2238. https://www.cnki.com.cn/Article/CJFDTOTAL-NHBH201910002.htm
Chen Y L, Weng L P, Ma J, et al. Review on the last ten years of research on source identification of heavy metal pollution in soils[J]. Journal of Agro-Environment Science, 2019, 38(10): 2219-2238. https://www.cnki.com.cn/Article/CJFDTOTAL-NHBH201910002.htm
[3] Liang J L, Huang X M, Yan J W, et al. A review of the formation of Cr(Ⅵ) via Cr(Ⅲ) oxidation in soils and groundwater[J]. Science of the Total Environment, 2021, 774: 145762. doi: 10.1016/j.scitotenv.2021.145762
[4] Zhang X W, Tong J X, Hu B X, et al. Adsorption and desorption for dynamics transport of hexavalent chromium (Cr(Ⅵ)) in soil column[J]. Environmental Science and Pollution Research, 2018, 25: 459-468. doi: 10.1007/s11356-017-0263-0
[5] Nagaraj P, Aradhana N, Shivakumar A, et al. Spe-ctrophotometric method for the determination of chromium (Ⅵ) in water samples[J]. Environmental Monitoring and Assessment, 2009, 157: 575-582. doi: 10.1007/s10661-008-0557-2
[6] Fikirte Z, Meareg A. Determination of the level of hexavalent, trivalent, and total chromium in the discharged effluent of Bahir Dar tannery using ICP-OES and UV-visible spectrometry[J]. Cogent Chemistry, 2018, 4(1): 1534566. doi: 10.1080/23312009.2018.1534566
[7] Miyake Y, Tokumura M, Iwazaki Y, et al. Determination of hexavalent chromium concentration in industrial waste incinerator stack gas by using a modified ion chromatography with post-column derivatization method[J]. Journal of Chromatography A, 2017, 1502: 24-29. doi: 10.1016/j.chroma.2017.04.046
[8] Borai E H, El-Sofany E A, Abdel-Halim A S. Speciation of hexavalent chromium in atmospheric particulate samples by selective extraction and ion chromatographic determination[J]. TrAC Trends in Analytical Chemistry, 2002, 21(11): 741-745. doi: 10.1016/S0165-9936(02)01102-0
[9] 林海兰, 谢沙, 文卓琼, 等. 碱消解-火焰原子吸收法测定土壤和固体废物中六价铬[J]. 分析试验室, 2017, 36(2): 198-202. https://www.cnki.com.cn/Article/CJFDTOTAL-FXSY201702017.htm
Lin H L, Xie S, Weng Z Q, et al. Determination of chromium (Ⅵ) in soil and solid waste by alkaline digestion-flame atomic absorption spectrometry[J]. Chinese Journal of Analysis Laboratory, 2017, 36(2): 198-202. https://www.cnki.com.cn/Article/CJFDTOTAL-FXSY201702017.htm
[10] Abkenar S D, Hosseini M, Dahaghin Z, et al. Speciation of chromium in water samples with homogeneous liquid-liquid extraction and determination by flame atomic absorption spectrometry[J]. Bulletin of the Korean Chemical Society, 2010, 31(10): 2813-2818. doi: 10.5012/bkcs.2010.31.10.2813
[11] Samira P, Mohammad B, Fatemeh Z, et al. Preconcentration and ultra-trace determination of hexavalent chromium ions using tailor-made polymer nanoparticles coupled with graphite furnace atomic absorption spectrometry: Ultrasonic assisted-dispersive solid-phase extraction[J]. New Journal of Chemistry, 2018, 42 (12): 10357-10365. doi: 10.1039/C8NJ01608A
[12] 炼晓璐, 魏洪敏, 甄长伟, 等. 碱消解-火焰原子吸收光谱法测定土壤中六价铬[J]. 中国无机分析化学, 2021, 11(3): 23-27. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX202103005.htm
Lian X L, Wei H M, Zhen C W, et al. Determination of hexavalent chromium in soil by alkali digestion flame atomic absorption spectrometry[J]. Chinese Jorunal of Inorganic Analytical Chemistry, 2021, 11(3): 23-27. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX202103005.htm
[13] 李强, 高存富, 曹莹, 等. 固体样品六价铬的检测比对和验证[J]. 环境工程, 2020, 38(6): 47-51. https://www.cnki.com.cn/Article/CJFDTOTAL-HJGC202006008.htm
Li Q, Gao C F, Cao Y, et al. Comparison and verification of hexavalent chromium detection in solid samples[J]. Environmental Engineering, 2020, 38(6): 47-51. https://www.cnki.com.cn/Article/CJFDTOTAL-HJGC202006008.htm
[14] 赵庆令, 李清彩, 谭现锋, 等. 微波碱性体系消解-电感耦合等离子体发射光谱法测定固体废物中的六价铬[J]. 岩矿测试, 2021, 40(1): 103-110. http://www.ykcs.ac.cn/cn/article/id/3e1263d3-6d6c-43f1-89da-736a5e4179b5
Zhao Q L, Li Q C, Tan X F, et al. Determination of hexavalent chromium in solid waste by inductively coupled plasma-optical emission spectrometry with microwave digestion[J]. Rock and Mineral Analysis, 2021, 40(1): 103-110. http://www.ykcs.ac.cn/cn/article/id/3e1263d3-6d6c-43f1-89da-736a5e4179b5
[15] 秦婷, 董宗凤, 吕晓华, 等. 碱消解-电感耦合等离子体发射光谱(ICP-OES)法测定土壤中六价铬[J]. 中国无机分析化学, 2019, 9(6): 10-13. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX201906003.htm
Qin T, Dong Z F, Lyh X H, et al. Determination of hexavalent chromium in soil by alkaline digestion-inductively coupled plasma optical emission spectrometry (ICP-OES)[J]. Chinese Jorunal of Inorganic Analytical Chemistry, 2019, 9(6): 10-13. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX201906003.htm
[16] 陈波, 胡兰. 电感耦合等离子体质谱法测定土壤样品中六价铬的前处理方法研究[J]. 理化检验(化学分册), 2021, 57(4): 358-361. https://www.cnki.com.cn/Article/CJFDTOTAL-LHJH202104016.htm
Chen B, Hu L. Study on pretreatment method for determination of hexavalent chromium in soil samples by inductively coupled plasma mass spectrometry[J]. Testing and Chemical Analysis (Part B: Chemical Analysis), 2021, 57(4): 358-361. https://www.cnki.com.cn/Article/CJFDTOTAL-LHJH202104016.htm
[17] Spanua D, Monticellia D, Binda G, et al. One-minute highly selective Cr(Ⅵ) determination at ultra-trace levels: An ICP-MS method based on the on-line trapping of Cr(Ⅲ)[J]. Journal of Hazardous Materials, 2021, 412: 125280. doi: 10.1016/j.jhazmat.2021.125280
[18] Barałkiewicz D, Pikosz B, Belter M, et al. Speciation analysis of chromium in drinking water samples by ion-pair reversed-phase HPLC-ICP-MS: Validation of the analytical method and evaluation of the uncertainty budget[J]. Accreditation and Quality Assurance, 2018, 18: 391-401.
[19] Christopher T M, Carleton R B, Ruth E W, et al. Modi-fications to EPA method 3060A to improve extraction of Cr(Ⅵ) from chromium ore processing residue-contaminated soils[J]. Environmental Science & Technology, 2017, 51: 11235-11243.
[20] Catalani S, Fostinelli J, Gilberti M E, et al. Application of a metal free high performance liquid chromatography with inductively coupled plasma mass spectrometry (HPLC-ICP-MS) for the determination of chromium species in drinking and tap water[J]. International Journal of Mass Spectrometry, 2015, 387: 31-37.
[21] 刘卫, 林建, 杨一, 等. 碱消解-离子色谱与电感耦合等离子体质谱(IC-ICP-MS)法测定土壤中的六价铬[J]. 中国无机分析化学, 2022, 12(1): 8-12. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX202201002.htm
Liu W, Lin J, Yang Y, et al. Determination of hexavalent chromium in soil by alkaline digestion-ion chromatography and inductively coupled plasma mass spectrometry (IC-ICP-MS)[J]. Chinese Journal of Inorganic Analytical Chemistry, 2022, 12(1): 8-12. https://www.cnki.com.cn/Article/CJFDTOTAL-WJFX202201002.htm
[22] Imanaka S, Hayashi H. Behavior of hexavalent chromium in the water supply system by IC-ICP-MS method[J]. Water Supply, 2013, 13(1): 96-103.
[23] Xiao W D, Zhang Y B, Li T Q, et al. Reduction kinetics of hexavalent chromium in soils and its correlation with soil properties[J]. Journal of Environmental Quality, 2012, 41(5): 1452-1458.
[24] Mädler S, Sun F, Tat C, et al. Trace-level analysis of hexavalent chromium in lake sediment samples using ion chromatography tandem mass spectrometry[J]. Journal of Environmental Protection, 2016, 7: 422-434.
[25] Huo D W, Kingston H M S. Correction of species transfor-mations in the analysis of Cr(Ⅵ) in solid environmental samples using speciated isotope dilution mass spectrometry[J]. Analytical Chemistry, 2000, 72: 5047-5054.
[26] Eary L E, Davis A. Geochemistry of an acidic chromium sulfate plume[J]. Applied Geochemistry, 2007, 22: 357-369.
[27] Novotnik B, Zuliani T, Šanar J, et al. The determination of Cr(Ⅵ) in corrosion protection coatings by speciated isotope dilution ICP-MS[J]. Journal of Analytical Atomic Spectrometry, 2012, 27: 1484-1493.
[28] 冷远鹏, 薛晓康, 章明洪. 土壤碱消解检测六价铬的铬还原问题及质控结果分析[J]. 安徽农业科学, 2019, 47(21): 206-208. https://www.cnki.com.cn/Article/CJFDTOTAL-AHNY201921064.htm
Leng Y P, Xue X K, Zhang M H. Determination of chromium reduction of hexavalent chromium by soil alkaline digestion and analysis of quality control results[J]. Journal of Anhui Agricultural Sciences, 2019, 47(21): 206-208. https://www.cnki.com.cn/Article/CJFDTOTAL-AHNY201921064.htm
[29] 刘海明, 武明丽, 成景特, 等. 酸溶分解-电感耦合等离子体质谱内标法测定地质样品中的痕量银[J]. 岩矿测试, 2021, 40(3): 444-450. http://www.ykcs.ac.cn/cn/article/doi/10.15898/j.cnki.11-2131/td.202002190018
Liu H M, Wu M L, Cheng J T, et al. Determination of trace silver in geological samples by inductively coupled plasma-mass spectrometry with acid decomposition and internal standard calibration[J]. Rock and Mineral Analysis, 2021, 40(3): 444-450. http://www.ykcs.ac.cn/cn/article/doi/10.15898/j.cnki.11-2131/td.202002190018
[30] Vanhaecke F, Vanhoe H, Dams R, et al. The use of internal standards in ICP-MS[J]. Talanta, 1992, 39(7): 737-742.
[31] 史凯, 朱建明, 吴广亮, 等. 地质样品中高精度铬同位素分析纯化技术研究进展[J]. 岩矿测试, 2019, 38(3): 341-353. http://www.ykcs.ac.cn/cn/article/doi/10.15898/j.cnki.11-2131/td.201805130059
Shi K, Zhu J M, Wu G L, et al. A review on the progress of purification techniques for high precision determination of Cr isotopes in geological samples[J]. Rock and Mineral Analysis, 2019, 38(3): 341-353. http://www.ykcs.ac.cn/cn/article/doi/10.15898/j.cnki.11-2131/td.201805130059
[32] Larsen K K, Wielandt D, Schiller M, et al. Chromatographic speciation of Cr(Ⅲ)-species, inter-species equilibrium isotope fractionation and improved chemical purification strategies for high-precision isotope analysis[J]. Journal of Chromatography A: Including Electrophoresis and Other Separation Methods, 2016, 1443: 162-174.
[33] 田晓芳, 高显超, 国静, 等. 过渡金属离子Mn(Ⅱ)和Fe(Ⅲ)对草酸还原Cr(Ⅵ)的催化作用[J]. 南京农业大学学报, 2009, 32(4): 160-164. https://www.cnki.com.cn/Article/CJFDTOTAL-NJNY200904031.htm
Tian X F, Gao X C, Guo J, et al. Catalytic role of Mn(Ⅱ) and Fe(Ⅲ) in the reduction of Cr(Ⅵ) by oxalic acid[J]. Journal of Nanjing Agricultural university, 2009, 32(4): 160-164. https://www.cnki.com.cn/Article/CJFDTOTAL-NJNY200904031.htm
[34] Vitale R J, Mussoline G R, Rinehimer K A, et al. Extrac-tion of sparingly soluble chromate from soils: Evaluation of methods and Eh-pH effects[J]. Environmental Science & Technology, 1997, 31(2): 390-394.
-



 下载:
下载: