REDOX ENVIRONMENT RESTORATION OF OCEAN WITH THE Fe/Mn RATIO OF CARBONATE ROCKS
-
摘要:
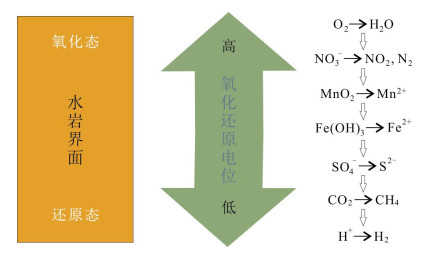
碳酸盐岩的Fe/Mn元素比值,作为一项新的地球化学指标,可以用于恢复海洋的氧化还原状态.在氧化条件下,Fe3+和Mn4+均不可溶,因此氧化海水中的溶解Fe和Mn的含量均很低.Fe3+和Mn4+在还原条件下可以被细菌还原为可溶的Fe2+和Mn2+,而氧化还原电位的计算表明,Mn4+的还原要早于Fe3+的还原,因此细菌的Mn还原过程发生在沉积物的更浅层.可溶的Fe2+和Mn2+向上扩散到海水中,替代碳酸盐岩晶格里的Ca2+,因此碳酸盐岩晶格中的Fe2+和Mn2+的含量受控于来自沉积物孔隙水的扩散,而后者又与水岩界面的氧化还原状态相关.因此可以预测,随着海水变得逐渐缺氧,碳酸盐岩中的Fe/Mn比值会逐渐增高.为了验证这一假说,我们分析了中元古代高于庄组白云岩的Fe/Mn比值.研究发现,几乎所有的样品的Fe/Mn比值介于20~30之间,显著高于泥盆纪末期深水碳酸盐岩和浅水台地碳酸盐岩的Fe/Mn比值.高于庄组碳酸盐岩高的Fe/Mn比值一方面可能指示了中元古代低的大气氧气浓度和海洋的广泛缺氧,也可能反映了白云岩形成于缺氧的沉积物空隙水里.
Abstract:As a new geochemical index, the Fe/Mn ratio of carbonate rocks can be used to restore the redox state of ocean. Both Fe3+ and Mn4+ are insoluble under the oxidation condition, so the dissolved Fe and Mn contents in oxidized seawater are very low. Fe3+ and Mn4+ can be reduced to soluble Fe2+ and Mn2+ by bacteria under the reduction condition, and the calculation of redox potential shows that the reduction of Mn4+ is earlier than that of Fe3+, thus the bacteria-induced Mn reduction process occurs in the shallower layer of sediments. The soluble Fe2+ and Mn2+ diffuse upward into seawater to replace the Ca2+ in lattice of carbonate rocks. The contents of Fe2+ and Mn2+ are controlled by the diffusion of porewater in sediments, while the latter is related to the redox state of water-rock interface. Therefore, it can be predicted that the Fe/Mn ratio in carbonate rocks would gradually increase as seawater becomes less oxygenated. The Fe/Mn ratio of dolomites in the Mesoproterozoic Gaoyuzhuang Formation is analyzed to test the hypothesis. It is found that the Fe/Mn ratio of almost all samples is 20-30, significantly higher than that of carbonate rocks in Late Devonian deep water and shallow water platform, which may indicate the low atmospheric oxygen concentration and wide oxygen depletion in ocean in Mesoproterozoic, and the dolomites were formed in the anoxic sediment porewater.
-
Key words:
- carbonate rock /
- ocean redox environment /
- geochemical index /
- Fe/Mn ratio
-

-
表 1 北票高于庄组碳酸盐岩Fe、Mn含量及Fe/Mn比值
Table 1. The Fe and Mn contents and Fe/Me ratios of the carbonate rock in Mesoproterozoic Gaoyuzhuang Formation
样品编号 碳酸盐组分含量/% Fe/10-6 Mn/10-6 Fe/Mn 样品编号 碳酸盐组分含量/% Fe/10-6 Mn/10-6 Fe/Mn 样品编号 碳酸盐组分含量/% Fe/10-6 Mn/10-6 Fe/Mn 18-BP-80 0.86 823.59 30.38 27.1 18-BP-53 0.93 993.78 45.27 22.0 18-BP-28 0.86 469.03 25.42 18.5 18-BP-79 0.99 875.05 30.17 29.0 18-BP-52 0.78 1010.01 38.17 26.5 18-BP-27 1.20 465.44 26.63 17.5 18-BP-78 0.83 651.04 33.46 19.5 18-BP-51 0.83 879.31 55.16 15.9 18-BP-26 0.86 465.46 35.64 13.1 18-BP-77+ 0.80 779.64 37.13 21.0 18-BP-50 0.78 984.80 43.67 22.6 18-BP-25 0.78 587.14 26.43 22.2 18-BP-77 0.96 877.09 30.91 28.4 18-BP-49 1.55 834.24 37.49 22.3 18-BP-24 1.01 724.06 73.38 9.9 18-BP-76 0.84 740.15 41.66 17.8 18-BP-48 0.69 848.80 37.17 22.8 18-BP-23 0.50 3642.06 38.95 93.5 18-BP-75 0.71 839.87 43.03 19.5 18-BP-47 0.89 871.67 34.65 25.2 18-BP-22 0.81 599.06 24.19 24.8 18-BP-74 0.54 572.88 36.57 15.7 18-BP-46 0.54 689.16 33.21 20.8 18-BP-21 1.05 647.17 26.04 24.9 18-BP-73 0.80 891.12 60.11 14.8 18-BP-45 0.85 803.32 43.53 18.5 18-BP-20 0.85 569.56 24.07 23.7 18-BP-72 0.75 1074.66 35.19 30.5 18-BP-44 0.80 785.05 36.51 21.5 18-BP-19 0.78 539.35 30.35 17.8 18-BP-71 0.86 624.85 26.63 23.5 18-BP-43 0.85 748.65 36.22 20.7 18-BP-18 0.99 491.37 28.53 17.2 18-BP-70 0.87 689.17 29.75 23.2 18-BP-42 1.04 815.94 38.25 21.3 18-BP-17 0.65 1018.11 37.82 26.9 18-BP-69 0.98 1118.12 48.82 22.9 18-BP-41 0.83 836.24 87.40 9.6 18-BP-16 1.24 760.22 36.20 21.0 18-BP-68 0.78 888.87 30.20 29.4 18-BP-40 0.49 822.45 41.75 19.7 18-BP-15 0.77 1012.29 37.26 27.2 18-BP-67 0.89 822.63 33.97 24.2 18-BP-39 0.56 1389.37 61.75 22.5 18-BP-14 0.94 457.89 24.95 18.4 18-BP-66 0.88 725.71 29.16 24.9 18-BP-38 0.79 1694.72 98.12 17.3 18-BP-13 0.53 957.41 51.75 18.5 18-BP-65 0.94 807.79 28.66 28.2 18-BP-37 0.88 945.82 33.09 28.6 18-BP-12 0.47 601.03 28.85 20.8 18-BP-64 0.85 785.68 29.16 26.9 18-BP-36 0.89 817.38 31.57 25.9 18-BP-11 0.81 522.35 26.52 19.7 18-BP-63 0.93 794.73 29.35 27.1 18-BP-35 0.98 861.73 33.37 25.8 18-BP-10 1.40 503.62 26.72 18.9 18-BP-62 0.83 527.45 24.29 21.7 18-BP-34Z 1.17 535.17 59.7 9.0 18-BP-09 1.34 725.19 32.17 22.5 18-BP-61 0.66 758.77 29.56 25.7 18-BP-34ZH 0.69 1138.90 75.02 15.2 18-BP-08 0.78 507.54 29.32 17.3 18-BP-60 0.68 897.37 45.23 19.8 18-BP-34QIAN 0.69 2917.70 81.34 35.9 18-BP-07 1.08 846.96 32.22 26.3 18-BP-59 0.27 1455.41 67.74 21.5 18-BP-33 0.86 813.80 36.01 22.6 18-BP-06 1.24 471.68 25.61 18.4 18-BP-58 0.81 910.91 31.10 29.3 18-BP-32 0.28 2356.56 106.71 22.1 18-BP-05 0.51 868.89 68.34 12.7 18-BP-57 1.06 929.69 31.25 29.8 18-BP-31 1.17 447.95 25.84 17.3 18-BP-04 1.01 446.97 29.63 15.1 18-BP-56 1.13 963.99 30.95 31.2 18-BP-30QIAN 0.66 744.20 34.00 21.9 18-BP-03 1.35 417.72 24.89 16.8 18-BP-55 0.86 886.72 35.16 25.2 18-BP-30 0.35 708.06 51.50 13.8 18-BP-02 0.91 533.85 22.56 23.7 18-BP-54 0.78 1141.86 47.58 24.0 18-BP-29 0.99 363.99 21.73 16.8 18-BP-01 1.14 587.71 27.26 21.6 测试单位:北京大学原德国斯派克ICP光谱分析合作实验室. -
[1] 常华进, 储雪蕾, 冯连君, 等.氧化还原敏感微量元素对古海洋沉积环境的指示意义[J].地质论评, 2009, 55(1):91-99. doi: 10.3321/j.issn:0371-5736.2009.01.011
[2] 许淑梅, 张晓东, 翟世奎, 等.海洋环境中氧化还原敏感性微量元素的地球化学行为及环境指示意义[J].海洋地质动态, 2007, 23(3):11-18. doi: 10.3969/j.issn.1009-2722.2007.03.002
[3] Wang Z, Guo W, Nie T, et al. Is seawater geochemical composition recorded in marine carbonate? Evidence from iron and manganese contents in Late Devonian carbonate rocks[J]. Acta Geochimica, 2019, 38(2):173-189. doi: 10.1007/s11631-018-00312-y
[4] 周炼, 苏洁, 黄俊华, 等.判识缺氧事件的地球化学新标志--钼同位素[J].中国科学:地球科学, 2011, 41(3):309-319. http://www.cnki.com.cn/Article/CJFDTotal-JDXK201103004.htm
[5] 刘春莲, 秦红, 车平, 等.广东三水盆地始新统心组生油岩元素地球化学特征及沉积环境[J].古地理学报, 2005, 7(1):125-136. doi: 10.3969/j.issn.1671-1505.2005.01.012
[6] Myers C R, Nealson K H. Bacterial manganese reduction and growth with manganese oxide as the sole electron acceptor[J]. Science, 1988, 240(4857):1319-1321. doi: 10.1126/science.240.4857.1319
[7] Nealson K H, Myers C R. Iron reduction by bacteria:A potential role in the genesis of banded iron formations[J]. American Journal of Science, 1990, 290:35-45.
[8] Thamdrup B. Bacterial manganese and iron reduction in aquatic sediments[C]//Advances in Microbial Ecology. Boston: Springer, 2000: 41-84.
[9] Canfield D E, Thamdrup B, Hansen J W. The anaerobic degradation of organic matter in Danish coastal sediments:Iron reduction, manganese reduction, and sulfate reduction[J]. Geochimica et Cosmochimica Acta, 1993, 57(16):3867-3883. doi: 10.1016/0016-7037(93)90340-3
-




 下载:
下载: