Formation mechanism of framboidal pyrite and its theory inversion of paleo-redox conditions
-
摘要:
研究目的 草莓状黄铁矿广泛存在于现代沉积物和沉积岩中,其成因机制总体上分为有机成因和无机成因两种,尽管两种机制均有理论与实验的支撑,但尚未建立一种具有普遍意义的形成机制。
研究方法 本文对目前草莓状黄铁矿的形成机理、氧化还原环境的应用及后期环境变化的影响进行了系统的综合研究。
研究结果 不同氧化-还原环境下形成的草莓状黄铁矿在粒径、形态以及硫同位素之间均存在较大的差异,可做为反演古氧化-还原环境的指标。草莓状黄铁矿的微晶尽管与粒径具有一定的正相关性,但是两者在形态演化序列、生长模式、聚集因素等方面与古氧化-还原环境的关系尚不清楚。仅凭草莓状黄铁矿粒径与铬还原法测定的硫同位素反演古氧化-还原环境存在一定的局限性,需要其他指标综合判定,尚需进一步开展草莓状黄铁矿原位硫同位素值与粒径对古氧化-还原环境反演的研究。后期氧化可使草莓状黄铁矿表面化学成分发生变化,但粒径分布依然具有古氧化-还原环境的指示意义。
结论 草莓状黄铁矿的实验模拟、理论体系和多学科交叉的研究中仍存在一些问题,尚需进一步研究。
Abstract:This paper is the result of mineral exploration engineering.
Objective Framboidal pyrite are widespread in modern sediments and sedimentary rocks, widely considered organic or inorganic genesis. Although both formation mechanisms have theoretical and experimental support, a formation mechanism with general significance has not yet been established well.
Methods This paper systematically and comprehensively studies the formation mechanism of framboidal pyrite, the application of redox conditions, and the influence of later environmental changes.
Results The size and texture of pyrite framboids and the sulfur isotopes between framboids have fluctuated with the oxygen level. Therefore, framboidal pyrite is used as a reconstruct paleoenvironment proxy commonly. Although the microcrystallines of framboidal pyrite are correlated to the particle size positively, their (Morphological evolution sequence), growth patterns, (aggregation factors), as well as the relationship with paleo-redox are still poorly understood. The redox condition inverse from particle sizes of pyrite framboids and chromium reduction-determined sulfur isotope has certain limitations. Therefore, a comprehensive analysis of redox indicators is expected, which requiring further studies on links between in-situ sulfur isotope and particle sizes of framboidal pyrite. Although the framboidal surface chemistry can be modified as changes in late oxidation conditions, the size distribution of framboidal pyrite is still meaningful as a redox indicator.
Conclusions In brief, experimental simulations, theoretical systems, and interdisciplinary studies on framboidal pyrite are still challenging and require further research.
-

-
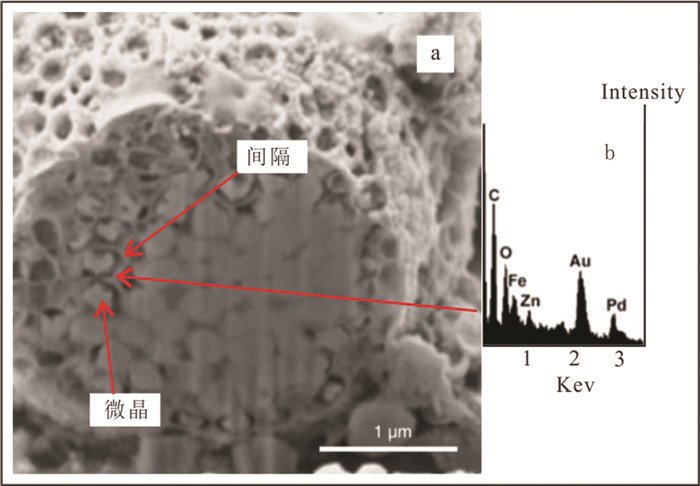
图 1 扫描电镜下草莓状黄铁矿外形及微晶结构(据Ohfuji et al., 2005)
Figure 1.
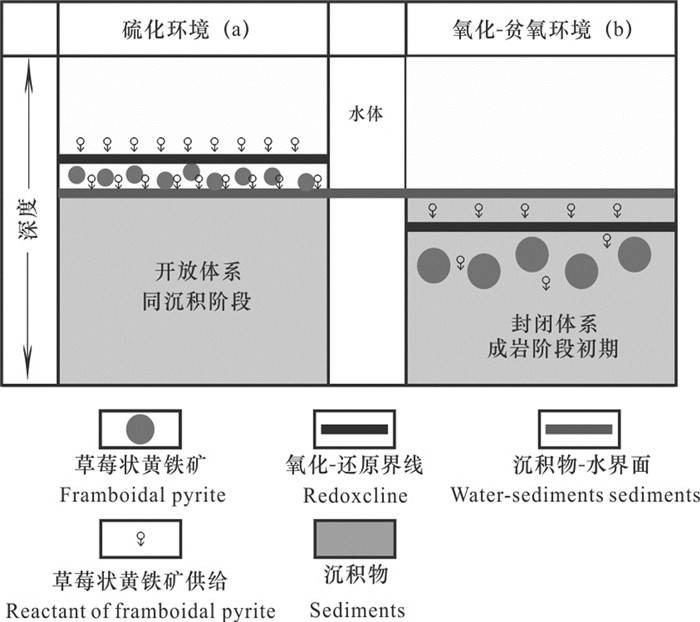
图 2 “草莓状黄铁矿有机成因-中空间隔模式”(据MacLean et al., 2008修改)
Figure 2.
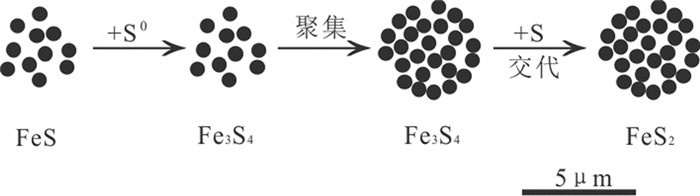
图 3 Wilkin和Barnes关于草莓状黄铁矿形成的模式图(据Wilkin et al., 1996;杨雪英等,2011)
Figure 3.
图 4 在25℃、0.1MPa的有限水体及沉积物环境下时间的对数与草莓状黄铁矿粒径关系图(据Rickard,2019)
Figure 4.
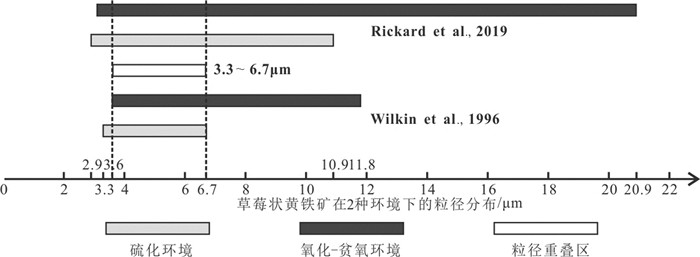
图 6 草莓状黄铁矿粒径在硫化环境和氧化-贫氧环境下的分布及明显的重叠分布(数据来源于Wilkin et al., 1996和Rickard, 2019)
Figure 6.
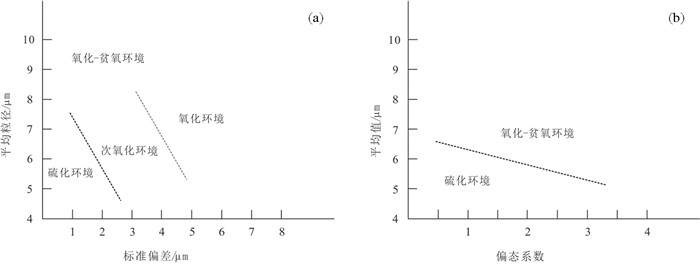
图 7 草莓状黄铁矿的平均粒径与标准偏差(a)、偏态系数(b)图解(据Wilkin et al., 1996;常华进等,2009)
Figure 7.
图 8 草莓状黄铁矿形态演化(据Merinero et al., 2008)
Figure 8.
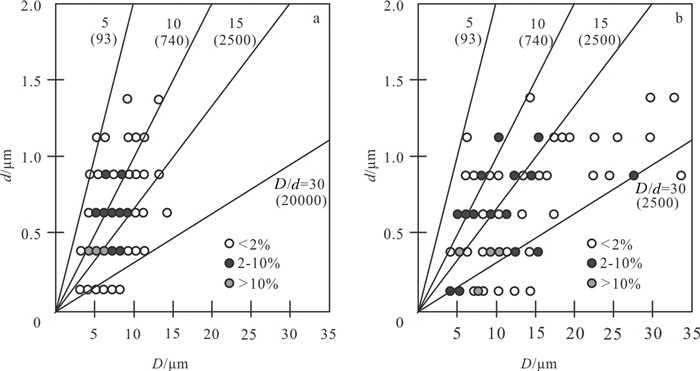
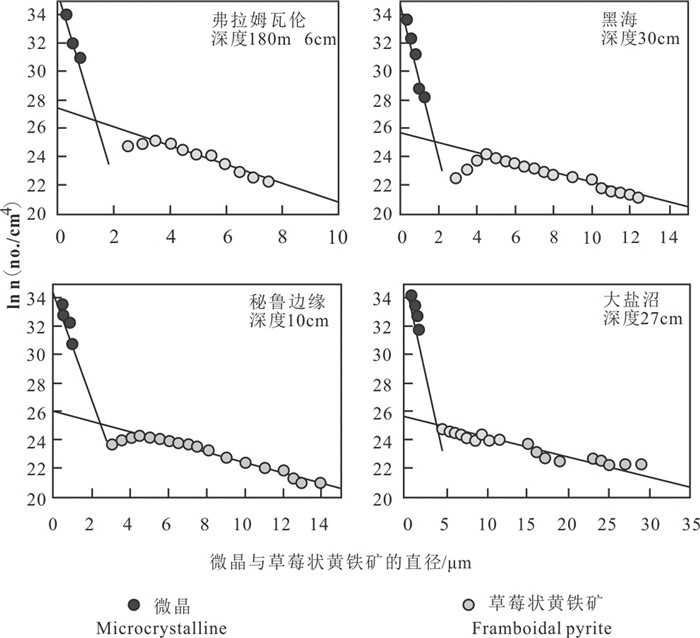
图 9 黑海沉积物(a)和大盐沼沉积物(b)中草莓状黄铁矿粒径(D)与微晶直径(d)的关系(据Wilkin et al., 1996)
Figure 9.
图 10 四个地点微晶与草莓状粒径分布图(据Wilkin et al., 1996)
Figure 10.
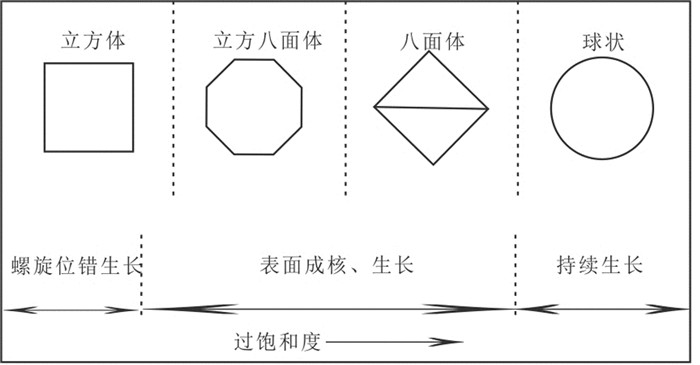
图 11 室温下一种黄铁矿微晶形态的演化和生长示意图(据Wang and Morse, 1996)
Figure 11.
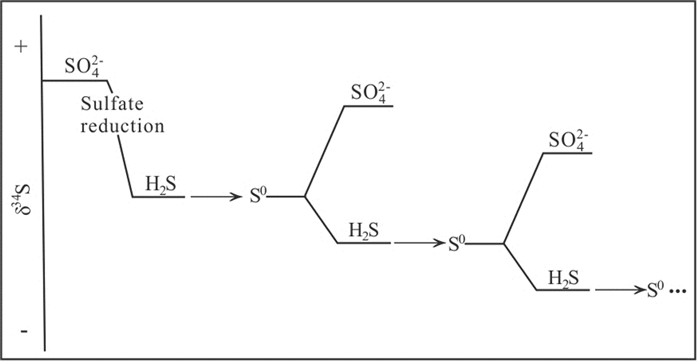
图 12 歧化反应过程中硫化物重复氧化为S0的模式图(据Canfield and Thamdrup, 1994)
Figure 12.
图 14 草莓状黄铁矿向自形黄铁矿演化的3种模式(据Sawlowicz,1993)
Figure 14.
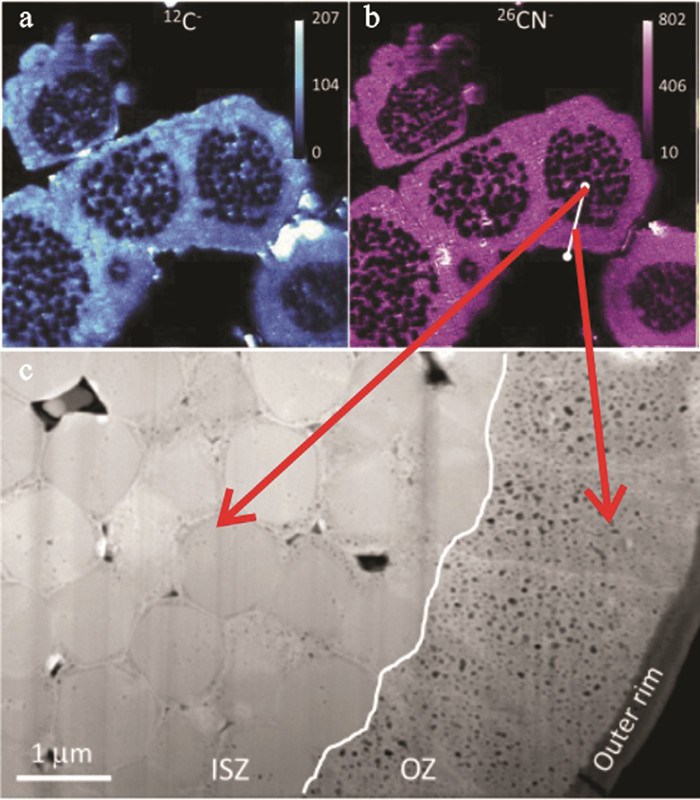
图 15 草莓状黄铁矿的二次生长对结构的改变(据Wacey et al., 2015)
Figure 15.
表 1 根据溶解氧划分的古氧化-还原环境(据Tyson and Pearson, 1991)
Table 1. Classification of paleo-redox conditions based on dissolved oxygen (after Tyson and Pearson, 1991)

表 2 草莓状黄铁矿粒径特征与古氧化-还原环境及沉积特征总结(据Bond and Wignall, 2010)
Table 2. Summary of characteristics used to define paleo-redox conditions during deposition(after Bond and Wignall, 2010)

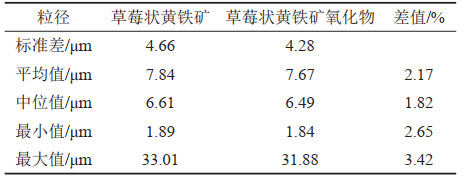
表 3 草莓状黄铁矿与草莓状黄铁矿氧化物粒径数据对比表(据黄元耕,2018)
Table 3. Comparison table of oxide particle size data of framboidal pyrite and framboidal pyrite (after Huang Yuangeng, 2018)
-
Berner R A. 1967. Thermodynamic stability of sedimentary iron sulfides[J]. American Journal of Science, 265: 773-785. doi: 10.2475/ajs.265.9.773
Berner R A. 1969. Migration of iron and sulfur within anaerobic sediments during early diagenesis[J]. American Journal of Science, 267(1): 19-42. doi: 10.2475/ajs.267.1.19
Berner R A, Raiswell R. 1983. Burial of organic carbon and pyrite sulfur in sediments over phanerozoic time A new theory[J]. Geochimica et Cosmochimica Acta, 47(5): 855-862. doi: 10.1016/0016-7037(83)90151-5
Berner R A. 1984. Sedimentary pyrite formation: An update[J]. Geochimica et Cosmochimica Acta, 48(4): 605-615. doi: 10.1016/0016-7037(84)90089-9
Bond D P G, Wignall P B W. 2010. Pyrite framboid study of marine Permian-Triassic boundary sections: A complex anoxic event and its relationship to contemporaneous mass extinction[J]. Geological Society of America Bulletin, 122(7-8): 1265-1279. doi: 10.1130/B30042.1
Bryant R N, Jones C, Raven M R, Owens J D, Fike D A. 2020. Shifting modes of iron sulfidization at the onset of OAE-2 drive regional shifts in pyrite δ34S records[J]. Chemical Geology, 553: 119808. doi: 10.1016/j.chemgeo.2020.119808
Butler I B, Rickard D. 2000. Framboidal pyrite formation via the oxidation of iron (II) monosulfide by hydrogen sulphide[J]. Geochimica et Cosmochimica Acta, 64(15): 2665-2672. doi: 10.1016/S0016-7037(00)00387-2
Canfield D E, Raiswell R, Westrich J T, Reaves C M, Berner R A. 1986. The use of chromium reduction in the analysis of reduced inorganic sulfur in sediments and shales[J]. Chemical Geology, 54(1/2): 149-155. https://www.sciencedirect.com/science/article/pii/0009254186900781
Canfield D E, Thamdrup B. 1994. The production of 34S-depleted sulfide during bacterial disproportionation of elemental sulfur[J]. Science, 266: 1973-1975. doi: 10.1126/science.11540246
Canfield D E, Habicht K S, Thamdrup B. 2000. The Archean sulfur cycle and the early history of atmospheric oxygen[J]. Science, 288: 658-661. doi: 10.1126/science.288.5466.658
Chang Huajin, Chu Xuelei, Feng Lianjun, Huang Jing. 2009. Framboidal pyrites in cherts of the Laobao Formation, South China: Evidence for anoxic deep ocean in the terminal Ediacaran[J]. Acta Petrologica Sinica, 25(4): 1001-1007(in Chinese with English abstract).
Chang Xiaolin, Huang Yuangen, Chen Zhongqiang, Hou Mingcai. 2020. The microscopic analysis of pyrite framboids and application in paleo-oceanography[J]. Acta Sedimentologica Sinica, 38(1): 150-165(in Chinese with English abstract).
Chen X, Rong J Y, Li A, Boucot J. 2004. Facies patterns and geography of the Yangtze region, South China, through the Ordovician and Silurian transition[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 204(3/4): 353-372. https://www.sciencedirect.com/science/article/pii/S0031018203007363
Cutter G A, Velinsky D J. 1988. Temporal variations of sedimentary sulfur in a Delaware salt marsh[J]. Marine Chemistry, 23: 311-327. doi: 10.1016/0304-4203(88)90101-6
Degens E T, Okada H, Honjo S, Hathaway J C. 1972. Microcrystalline sphalerite in resin globules suspended in Lake Kivu, East Africa[J]. Mineralium Deposita, 7(1): 1-12.
Donald R, Southam G. 1999. Low temperature anaerobic bacterial diagenesis of ferrous monosulfide to pyrite[J]. Geochimica et Cosmochimica Acta, 63: 2019-2023. doi: 10.1016/S0016-7037(99)00140-4
England B M, Ostwald J. 1993. Framboid-derived structures in some Tasman fold belt base-metal sulphide deposits, New South Wales, Australia[J]. Ore Geology Reviews, 7(5): 381-412. doi: 10.1016/0169-1368(93)90002-G
Fan Hongrui, Li Xinghui, Zuo Yabin, Chen Lei, Liu Wei, Hu Fangfang, Feng Kai. 2018. In-situ LA-(MC)-ICPMS and (Nano) SIMS trace elements and sulfur isotope analyses on sulfides and application to confine metallogenic process of ore deposit[J]. Acta Petrologica Sinica, 34(12): 3479-3496. (in Chinese with English abstract).
Farina M, Esquivel D, Henrique G P, Barros L D. 1990. Magnetic iron-sulphur crystals from a magnetotactic microorganism[J]. Nature, 343: 256-258. doi: 10.1038/343256a0
Farquhar J, Bao H, Thiemens M. 2000. Atmospheric influence of Earth's earliest sulfur cycle[J]. Science, 289(5480): 756-759. doi: 10.1126/science.289.5480.756
Farquhar J, Savarino J, Airieau S, Mark H, Thiemens M H. 2001. Observation of wavelength-sensitive mass-independent sulfur isotope effects during SO2 photolysis: Implications for the early atmosphere[J]. Journal of Geophysical Research: Planets, 106(E12): 32829-32839. doi: 10.1029/2000JE001437
Farrand M. 1970. Framboidal sulphides precipitated synthetically[J]. Mineralium Deposita, 5(3): 237-247. https://link.springer.com/article/10.1007%2FBF00201990
Gao Yongwei, Wang Zhihua, Li Weiliang, Zhang Zhenliang. 2019. A review of pyrite mineralogy research in hydrothermal gold deposits[J]. Northwestern Geology, 52(3): 58-69(in Chinese with English abstract).
Goldhaber M B, Kaplan I R. 1975. Controls and consequences of sulfate reduction rates in recent marine sediments[J]. Soil Sci., 119: 42-55. doi: 10.1097/00010694-197501000-00008
Gomes M L, Fike D A, Bergmann K D, Jones C, Knoll A H. 2018. Environmental insights from high-resolution (SIMS) sulfur isotope analyses of sulfides in Proterozoic microbialites with diverse mat textures[J]. Geobiology, 16(1): 17-34. doi: 10.1111/gbi.12265
Habicht K S, Canfield D E. 1997. Sulfur isotope fractionation during bacterial sulfate reduction in organic-rich sediments[J]. Geochimica et Cosmochimica Acta, 61(24): 5351-5361. doi: 10.1016/S0016-7037(97)00311-6
Habicht K S, Gade M, Thamdrup B, Berg P, Canfield D E. 2002. Calibration of sulfate levels in the archean ocean[J]. Science, 298(5602): 2372-2374. doi: 10.1126/science.1078265
Huang Fei, Gao Shang, Chen Lei, Su Limin, Li Yongli, Meng Lin, Liu Kaijun, Chai Chenwei, Qi Xinyi. 2020. Micro-texture and in situ sulfur isotope of pyrite from the Baiyunpu Pb-Zn deposit in central Hunan, South China: Implications for the growth mechanism of colloform pyrite aggregates[J]. Journal of Asian Earth Science, 193(15).
Hartman P, Perdok W G. 1955. On the relations between structure and morphology of crystal1[J]. Acta Crystallographica(Section A), 8(1): 49-52. doi: 10.1107/S0365110X55000121
Horiuchi S, Wade H, Moori T. 1974. Morphology and imperfection of hydrothermally synthesized greigite (Fe3S4)[J]. Journal of Crystal Growth, 24: 624-626.
Hu Yongliang, Wang wei, Zhou Chuanming. 2020. Morphologic and Isotopic Characteristics of Sedimentary Pyrite: A case study from deepwater facies, Ediacaran Lantian Formation in South China[J]. Acta Sedimentologica Sinica, 38(1): 138-149(in Chinese with English abstract).
Huang Y, Chen Z, Algeo T J, Zhao L, Baud A, Bhat G M. 2019. Two-stage marine anoxia and biotic response during the Permian-Triassic transition in Kashmir, northern India: Pyrite framboid evidence[J]. Global and Planetary Change, 172: 124-139. doi: 10.1016/j.gloplacha.2018.10.002
Kalliokoski J, Cathles L. 1969. Morphology, mode of formation, and diagenetic changes in framboids[J]. Bulletin of the Geological Society of Finland, 41: 125-133. doi: 10.17741/bgsf/41.014
Konhauser K O. 1997. Bacterialiron biomineralisation in nature[J]. FEMS Micrology Review, 20: 315-326. doi: 10.1111/j.1574-6976.1997.tb00317.x
Lin M, Huang F, Wang X Q, Gao W Y, Zhang B M, Song D, Li G L, Zhang B Y. 2020. An experimental study of the morphological evolution of pyrite under hydrothermal conditions and its implications[J]. Journal of Geochemical Exploration, 219. https://www.sciencedirect.com/science/article/pii/S0375674219306533
Lin Z, Sun X, Peckmann J, Lu Y, Xu L, Strauss H, Zhou H, Gong J, Lu H, Teichert B M A. 2016. How sulfate-driven anaerobic oxidation of methane affects the sulfur isotopic composition of pyrite: A SIMS study from the South China Sea[J]. Chemical Geology, 440: 26-41. doi: 10.1016/j.chemgeo.2016.07.007
Liu Bin, Chen Weifeng, Fang Qichun, Tang Xiangsheng, Mao Yufeng, Sun Liqiang, Gao Shuang, Yan Yongjie, Wei Xin, Ling Hongfei. 2020. Study on in-situ sulfur isotope compositions of sulfides: Implication for the source of Pb-Zn mineralized body of Niutoushan in the Xiangshan Area[J]. Earth Science, 45(2): 389-399(in Chinese with English abstract). https://www.researchgate.net/publication/250130914_Sulfur_Isotope_Geochemistry_of_Sulfide_Minerals
Liu Dameng, Yang Qi, Zhou Chunguang, Tang Dazhen, Kang Xidong. 1999. Occurrence and geological gensis of pyrite in Late Paleozoic coals in north china[J]. Geochimica, 28(4): 340-350(in Chinese with English abstract).
Love L G. 1957. Mircro-organisms and the presence of syngenetic pyrite[J]. Quarterly Journal of the Geological Society, 113: 429-440. doi: 10.1144/GSL.JGS.1957.113.01-04.18
Love L G, Amstutz G C. 1966. Review of microscopic pyrite from the Devonian chattanooga shale and rammelserg banderz[J]. Fortschr Mineral, 43: 273-309.
Love L G, Brockley H. 1973. Peripheral radial texture in framboids of polyframboidal pyrite[J]. Fortschr. Miner, 50: 264-269.
Lowenstam H. 1981. Minerals formed by organisms[J]. Science, 211(4487): 1126-1131. doi: 10.1126/science.7008198
MacLean L C, Tyliszczak T, Gilbert P U, Zhou D, Pray T J, Onstott T C, Southam G. 2008. A high-resolution chemical and structural study of framboidal pyrite formed within a low-temperature bacterial biofilm[J]. Geobiology, 6(5): 471-480. doi: 10.1111/j.1472-4669.2008.00174.x
Magnall J M, Gleeson S A, Stern R A, Newton R J, Poulton S W, Paradis S. 2016. Open system sulphate reduction in a diagenetic environment-Isotopic analysis of barite (δ34S and δ18O) and pyrite (δ34S) from the Tom and Jason Late Devonian Zn-Pb-Ba deposits, Selwyn Basin, Canada[J]. Geochimica et Cosmochimica Acta, 180: 146-163. doi: 10.1016/j.gca.2016.02.015
Merinero R, Lunar R, Frias J M, Somoza L, Diaz-del-Rio V d. 2008. Iron oxyhydroxide and sulphide mineralization in hydrocarbon seep-related carbonate submarine chimneys, Gulf of Cadiz (SW Iberian Peninsula)[J]. Marine and Petroleum Geology, 25(8): 706-713. doi: 10.1016/j.marpetgeo.2008.03.005
Meyer K M, Alonso R M, Morrissey S, Jones C. 2019. Sulfur Isotope Measurements of Framboidal Pyrites from the Sediments and Water Column of a Stratified Euxinic Lake[C]//American Geophysical Union.
Moore R A, Lieberman B S. 2009. Preservation of early and Middle Cambrian soft-bodied arthropods from the Pioche shale, Nevada, USA[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 277(1/2): 57-62. https://www.sciencedirect.com/science/article/pii/S0031018209000856
Morse J, Wang W Q. 1997. Pyrite formation under conditions approximating those in anoxic sediments: II. Influence of precursor iron minerals and organic matter[J]. Marine Chemistry, 57: 187-193. doi: 10.1016/S0304-4203(97)00050-9
Muramoto J Honjo A S B F, Hay B J, Howarth R W, Cisne J L. 1991. Sulfur, iron and organic carbon fluxes in the Black Sea: Sulfur isotopic evidence for origin of sulfur fluxes[J]. Deep Sea Research Part A. Oceanographic Research Papers, 38: S1151-S1187. doi: 10.1016/S0198-0149(10)80029-9
Nozaki T, Nagase T, Ushikubo T, Shimizu K, Ishibashi J i, the D/V Chikyu Expedition 909 Scientists. 2020. Microbial sulfate reduction plays an important role at the initial stage of subseafloor sulfide mineralization[J]. Geology, 49(2): DOI:10.1130/G47943.1.
Ohfuji H D Rickard. 2005. Experimental syntheses of framboids-a review[J]. Earth-Science Reviews, 71(3/4): 147-170. https://www.sciencedirect.com/science/article/pii/S001282520500019X
Popa R, Kinkle B K, Badescu A. 2004. Pyrite framboids as biomarkers for Iron-Sulfur Systems[J]. Geomicrobiology, 21(3): (193-206). doi: 10.1080/01490450490275497
Raiswell R. 1982. Pyrite, texture isotopic composition and the availability of iron[J]. American Journal of Science, 282: 1244-1263. doi: 10.2475/ajs.282.8.1244
Raiswell R, Berner R A. 1985. Pyrite formation in euxinic and semi-euxinic sediments[J]. American Journal of Science, 285(8): 710-724. doi: 10.2475/ajs.285.8.710
Randolf A D, Larson W A. 1971. Theory of Particulate Processes. Analysis and Techniques of Continuous Crystallization[M]. Academic Press, New York and London.
Rickard D. 2019. How long does it take a pyrite framboid to form[J]. Earth & Planetary Science Letters, 513: 64-68. https://www.sciencedirect.com/science/article/pii/S0012821X19301165
Rickard D. 2019. Sedimentary pyrite framboid size-frequency distributions: A meta-analysis[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 522: 62-75. doi: 10.1016/j.palaeo.2019.03.010
Rickard D T. 1970. The origin of framboids[J]. Lithos, 3(3): 269-293. doi: 10.1016/0024-4937(70)90079-4
Rust G W. 1935. Colloidal primary copper ores at Cornwall mines, Southeastern Missouri[J]. The Journal of Geology, 43(4): 398-426. doi: 10.1086/624318
Sawlowicz Z. 1993. Pyrite framboids and their development: A new conceptual mechanism[J]. Geologische Rundschau, 82(1): 148-156. doi: 10.1007/BF00563277
Sawlowicz Z. 1987. Framboidal pyrite from the metamorphic Radzimowice Schists of Stara Gora(Lower Silesia, Poland)[J]. Mineral Polon, 18: 57-67.
Steinike K. 1963. A further remark on biogenic sulfides: Inorganic pyrite spheres[J]. Econimic Geology, 58(6): 998-1000. doi: 10.2113/gsecongeo.58.6.998
Sun G T, Zeng Q D, Zhou L L, Wang Y B, Chen P W. 2020. Trace element contents and in situ sulfur isotope analyses of pyrite in the Baiyun gold deposit, NE China: Implication for the genesis of intrusion-related gold deposits[J]. Ore Geology Reviews, 11810.10161j. oregeorev. 2020.103330. https://www.sciencedirect.com/science/article/pii/S0169136819306948
Sweeney R E, Kaplan I R. 1973. Pyrite framboid formation laboratory synthesis and marine sediments[J]. Economic Geology, 68(5): 618-634. doi: 10.2113/gsecongeo.68.5.618
Tauson V L, Abramovich M G, Akimov V V, Scherbakov V A. 1993. Thermodynamics of real mineral crystals: Equilibriumcrystal shape and phase size effect[J]. Geochimica et Cosmochimica Acta, 57: 815-821. doi: 10.1016/0016-7037(93)90170-2
Taylor G R. 1982. A mechanism for framboid formation as illustrated by a volcanic exhalative sediment[J]. Mineralium Deposita, 17(1): 23-36. https://link.springer.com/article/10.1007/BF00206374
Tyson R V, Pearson T H. 1991. Modern and ancient continental shelf anoxia: An overview[J]. Geol. Soc. Spec. Pub., 58: 1-24. doi: 10.1144/GSL.SP.1991.058.01.01
Wacey D, Kilburn M R, Saunders M, Cliff J B, Kong C, Liu A G, Matthews J J, Brasier M D. 2015. Uncovering framboidal pyrite biogenicity using nano-scale C/Norg mapping[J]. Geology, 43(1): 27-30. doi: 10.1130/G36048.1
Wang Q, Morse J W. 1996. Pyrite formation under conditions approximating those in anoxic sediments I. Pathway and morphology[J]. Marine Chemistry, 52(2): 99-121. doi: 10.1016/0304-4203(95)00082-8
Wei H Y, Wei X M, Qiu Z, Song H Y, Guo S. 2016. Redox conditions across the G-L boundary in South China: Evidence from pyrite morphology and sulfur isotopic compositions[J]. Chemical Geology, 440: 1-14. doi: 10.1016/j.chemgeo.2016.07.009
Wignall P B, Newton R. 1998. Pyrite framboid diameter as a measure of oxygen deficiency in ancient mudrocks[J]. American Journal of Science, 298(7): 537-552. doi: 10.2475/ajs.298.7.537
Wilkin R T. 1995. Size Distribution in Sediments, Synthesis, and Formation Mechanism of Framboidal[D]. PhD. Dissertation. The Pennsylvania State Universty,
Wilkin R T, Barnes H L, Brantly S L. 1996. The size distribution of framboidal pyrite in modern sediments: An indicator of redox conditions[J]. Geochimica et Cosmochimica Acta, 60(20): 3897-3912. doi: 10.1016/0016-7037(96)00209-8
Wilkin R T, Barnes H L. 1997a. Formation processes of framboidal pyrite[J]. Geochimica et Cosmochimica Acta, 61(2): 323-339. doi: 10.1016/S0016-7037(96)00320-1
Xiao Fan, Ban Yizhong, Fan Feipeng, Xu Naicen, Mao Guangwu, Li Fengchun. 2020. Research on zircon U-Pb, S-Pb isotopes and trace elements of pyrite from the Dongji Au(Ag) deposit in Zhenghe County, Fujian Province[J]. Geology in China, 47(2): 375-393(in Chinese with English abstract).
Xu Nan, Wu Cailai, Li Shengrong, Xue Boqiang, He Xiang, Yu Yanlong, Liu Junzhuang. 2020. LA-ICP-MS in situ analyses of the pyrites in Dongyang gold deposit, Southeast China: Implications to the gold mineralization[J]. China Geology, 3: 230-246. https://www.sciencedirect.com/science/article/pii/S2096519220300744
Yan D, Chen D, Wang Q, Wang J. 2012. Predominance of stratified anoxic Yangtze Sea interrupted by short-term oxygenation during the Ordo-Silurian transition[J]. Chemical Geology, 291: 69-78. doi: 10.1016/j.chemgeo.2011.09.015
Yang R, He S, Wang X, Hu Q, Hu D, Yi J, Widory D. 2016. Paleo-ocean redox environments of the Upper Ordovician Wufeng and the first member in lower Silurian Longmaxi formations in the Jiaoshiba area, Sichuan Basin[J]. Canadian Journal of Earth Sciences, 53(4): 426-440. doi: 10.1139/cjes-2015-0210
Yang Xueying, Gong Yiming. 2011. Pyrite framboid: Indicator of environments and life[J]. Earth Science, 36(4): 643-658(in Chinese with English abstract).
Zhang Wei, Liu Congqiang, Liang Xiaobing. 2007. Biological function in sulfur isotope fractionation and environmental effect[J]. Acta Geochimica, 35(3): 223-227 (in Chinese with English abstract).
Zheng J H, Shen P, Li C H. 2020. Ore genesis of Axi post-collisional epithermal gold deposit, western Tianshan, NW China: Constraints from U-Pb dating, Hf isotopes, and pyrite in situ sulfur isotopes[J]. Ore Geology Review, 117 DOI:10. 1016/j. orgrorev. 2019. 103290.
Zhou Lingli, Zeng Qingdong, Sun Guotao, Duan Xiaoxia. 2019. Laser Ablation-Inductively Coupled Plasma Mass Spectrometry (LA-ICPMS) elemental mapping and its applications in ore geology[J]. Acta Petrologica Sinica, 35(7): 1964-1978 (in Chinese with English abstract). doi: 10.18654/1000-0569/2019.07.02
Zhu Xiangkun, Wang Yue, Yan Bin, Li Jin, Dong Aiguo, Li Zhihong, Sun Jian. 2013. Developments of non-traditional stable isotope geochemistry[J]. Bulletin of Mineralogy, Petrology and Geochemistry, 32(6): 651-688 (in Chinese with English abstract). https://www.researchgate.net/publication/273766379_Developments_of_Non-Traditional_Stable_Isotope_Geochemistry
Zou C N, Zhen Q, W H Y, Dong D Z, Lu B. 2018. Euxinia caused the Late Ordovician extinction Evidence from pyrite morphology and pyritic sulfur isotopic composition in the Yangtze area, South China[J]. Palaeogeography, Palaeoclimatology, Palaeoecology, 511: 1-11. doi: 10.1016/j.palaeo.2017.11.033
常华进, 储雪蕾, 冯连君, 黄晶. 2009. 华南老堡组硅质岩中草莓状黄铁矿-埃迪卡拉纪末期深海缺氧的证据[J]. 岩石学报, 25(4): 1001-1007. https://d.wanfangdata.com.cn/periodical/ysxb98200904024
常晓琳, 黄元耕, 陈中强, 侯明才. 2020. 沉积地层中草莓状黄铁矿分析方法及其在古海洋学上的应用[J]. 沉积学报, 1: 150-165. http://www.cnki.com.cn/Article/CJFDTotal-CJXB202001013.htm
范宏瑞, 李兴辉, 左亚彬, 陈蕾, 刘尚, 胡芳芳, 冯凯. 2018. LA-(MC)-ICPMS和(Nano)SIMS硫化物微量元素和硫同位素原位分析与矿床形成的精细过程[J]. 岩石学报, 34(12): 3479-3496. http://www.cnki.com.cn/Article/CJFDTotal-YSXB201812002.htm
胡永亮, 王伟, 周传明. 2020. 沉积地层中的黄铁矿形态及同位素特征初探——以华南埃迪卡拉纪深水相地层为例[J]. 沉积学报, 38(1): 138-149. http://www.cnki.com.cn/Article/CJFDTotal-CJXB202001012.htm
黄元耕. 2018. 华南及新疆地区二叠纪至三叠纪海洋、陆地古群落模拟及海洋氧化还原环境变化研究[D]. 中国地质大学(武汉).
刘斌, 陈卫锋, 方启春, 唐湘生, 毛玉锋, 孙立强, 高爽, 严永杰, 魏欣, 凌洪飞. 2020. 相山西部牛头山铅锌矿化体成矿物质来源: 原位硫同位素的制约[J]. 地球科学, 45(2): 389-399. https://www.cnki.com.cn/Article/CJFDTOTAL-DQKX202002004.htm
刘大猛, 杨起, 周春光, 康西栋. 1999. 华北晚古生代煤中黄铁矿赋存特征与地质成因研究[J]. 地球化学, 28(4): 340-351. doi: 10.3321/j.issn:0379-1726.1999.04.004
韦雪梅. 2017. 广西蓬莱滩GSSP剖面G-L界线黄铁矿形态特征及其氧化还原意义[D]. 东华理工大学.
肖凡, 班宜忠, 范飞鹏, 许乃岑, 毛光武, 李凤春. 2020. 福建政和县东际金(银)矿床黄铁矿微量元素和硫-铅同位素及锆石年龄研究[J]. 中国地质, 47(2): 375-393. http://geochina.cgs.gov.cn/geochina/ch/reader/view_abstract.aspx?file_no=20200208&flag=1
杨雪英, 龚一鸣. 2011. 莓状黄铁矿: 环境与生命的示踪计[J]. 地球科学, 36(4): 643-658. http://www.cnki.com.cn/Article/CJFDTotal-DQKX201104004.htm
张伟, 刘丛强, 梁小兵. 2007. 硫同位素分馏中的生物作用及其环境效应[J]. 地球与环境, 35(3): 223-227. https://www.cnki.com.cn/Article/CJFDTOTAL-DZDQ200703004.htm
周伶俐, 曾庆栋, 孙国涛, 段晓侠. 2019. LA-ICPMS原位微区面扫描分析技术及其矿床学应用实例[J]. 岩石学报, 35(7): 1964-1978. http://www.cnki.com.cn/Article/CJFDTotal-YSXB201907002.htm
朱祥坤, 王跃, 闫斌, 李津, 董爱国, 李志红, 孙剑. 2013. 非传统稳定同位素地球化学的创建与发展[J]. 矿物岩石地球化学通报, 32: 651-688. http://www.cnki.com.cn/Article/CJFDTotal-KYDH201306002.htm
-




 下载:
下载: