Study on Vanadium Extraction from Leachate of Low Grade Vanadium Ore by Ion Exchange Method
-
摘要:
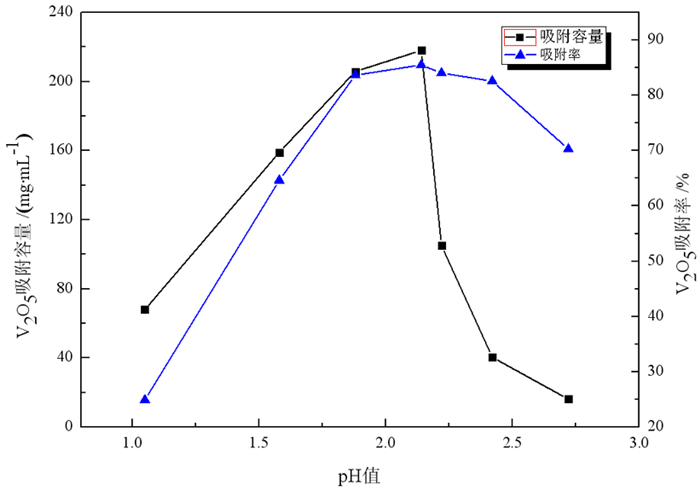
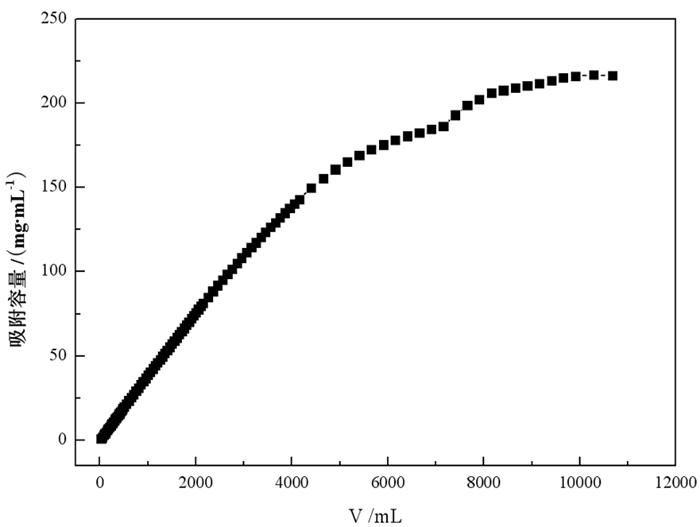
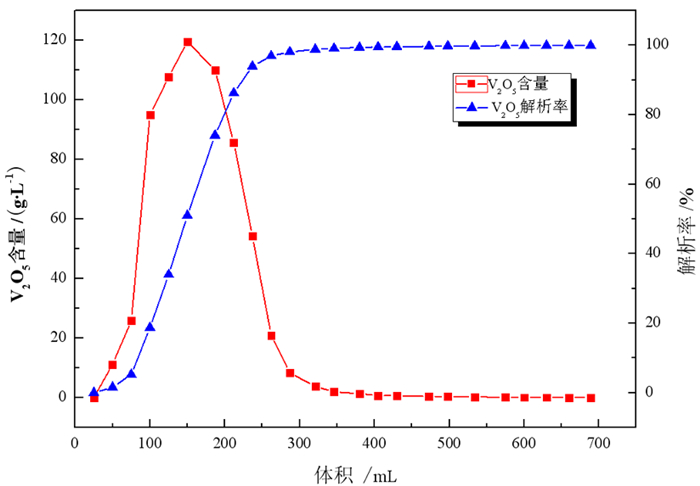
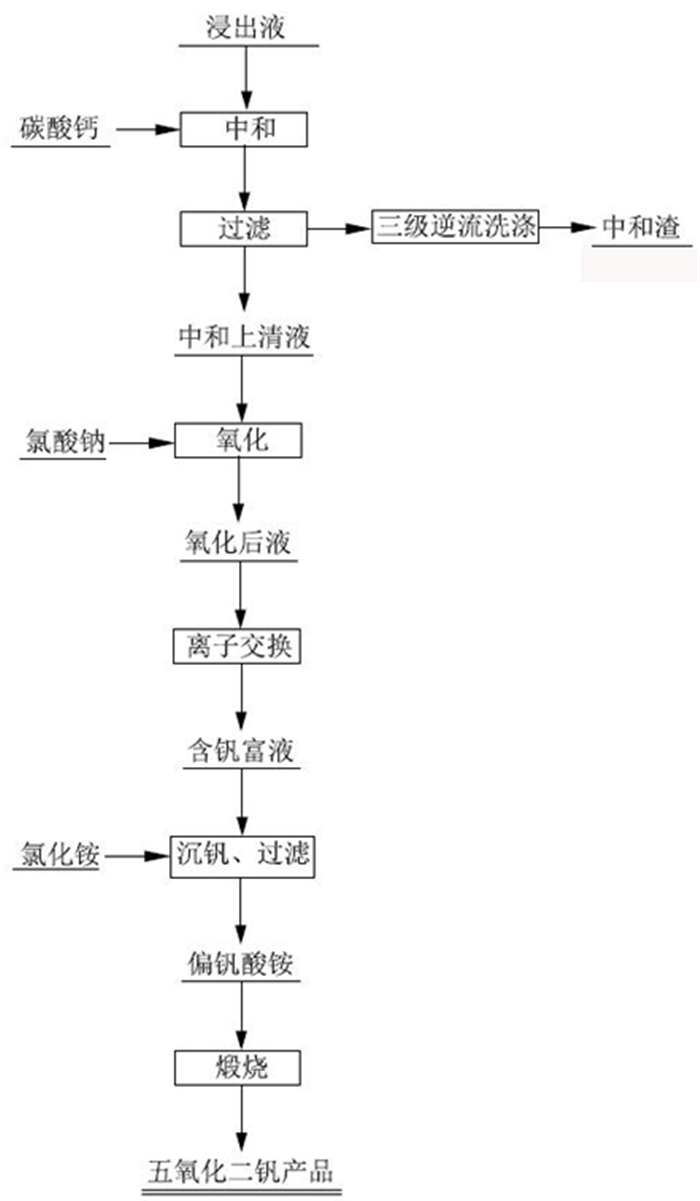
采用离子交换法对石煤钒矿通过拌酸保温熟化—堆浸所得浸出液进行了提钒工艺研究, 主要考察离子交换法提钒工艺, 树脂类型、溶液pH值、动态吸附对钒吸附率的影响。试验结果表明, 含钒浸出液经中和—氧化后调节pH=1.88得到交换前液, 优选吸附性能较好的阴离子交换树脂D201, 进行动态吸附, V2O5饱和吸附容量217.66 mg/mL湿树脂; 以4% NaOH+4% NaCl配比的解吸剂对D201进行动态解吸, 解吸液中V2O5含量最高达到119.49 g/L, 在解吸液体积(mL)与湿树脂体积(mL)比为3.7时, 树脂解吸率已超过99%;所得解吸液经酸性铵盐一步沉钒法制备得到品位超过98%的高纯V2O5产品。本试验提供了一种操作方便、成本低廉的提钒工艺, 树脂D201具有对钒吸附容量大、吸附率高、处理量大等优势, 富钒解吸液无需净化处理, 利用铵盐一步沉钒法最终得到合格产品, 并且避免了萃取工艺带来的废水处理难度大的问题, 工艺适应性强。
-
关键词:
- 石煤钒矿 /
- 离子交换法 /
- 阴离子交换树脂D201
Abstract:The ion exchange method was used to study the extraction process of vanadium from the leaching solution of stone coal vanadium ore obtained by mixing acid, heat preservation, maturation and heap leaching. The influence of resin type, pH value of solution and dynamic adsorption on vanadium adsorption rate was mainly investigated. The experimental results showed that the vanadium-containing leaching solution was neutralized and oxidized to obtain the pre-exchange solution by adjusting pH=1.88, and the anion exchange resin D201 with better adsorption performance was selected for dynamic adsorption. The saturated adsorption capacity of V2O5 was 217.66 mg/mL wet resin. The dynamic desorption of D201 was carried out with 4%NaOH + 4%NaCl. The maximum content of V2O5 in the desorption solution was 119.49 g/L. When the volume ratio of desorption solution (mL) to wet resin (mL) was 3.7, the desorption rate of resin was over 99%. The obtained analytical solution was prepared by one-step precipitation of vanadium with acidic ammonium salt to obtain high-purity V2O5 products with a grade of over 98%. This experiment provides a convenient operation and low cost of vanadium extraction process. The resin D201 has the advantages of large adsorption capacity, high adsorption rate and large treatment capacity for vanadium. The vanadium-rich analytical solution does not need purification treatment. The ammonium salt one-step precipitation method is used to obtain qualified products, and the difficulty of wastewater treatment caused by the extraction process is avoided. The process is highly adaptable.
-
Key words:
- stone coal vanadium ore /
- ion exchange method /
- anion exchange resin D201
-

-
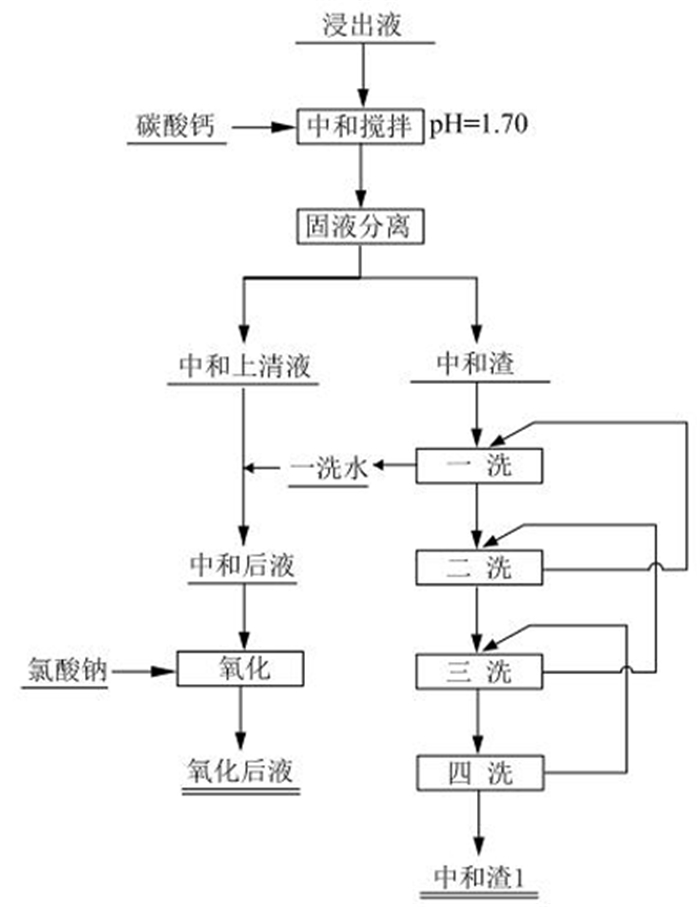
表 1 浸出液成分分析
Table 1. Component analysis of leaching solution
成分 V2O5 TFe Al2O3 Na2O K2O 含量/(g·L-1) 2.50 7.63 7.97 0.096 1.51 成分 SiO2 U Hg Ti CaO 含量/(mg·L-1) 0.12 5.76 0.002 37.1 0.16 表 2 中和—氧化试验结果
Table 2. Neutralization-oxidation experimental results
溶液类型 pH 氧化还原电位/mV V/mL V2O5含量/(g·L-1) V2O5分布率/% 浸出液 0.72 476 1000 3.43 / 中和上清液 1.84 / 530 2.97 49.18 一洗水 2.48 / 500 1.26 19.68 二洗水 2.05 / 555 0.94 16.30 三洗水 2.70 / 510 0.37 5.90 四洗水 2.84 / 890 0.09 2.50 中和渣 / / 54.17 g 0.38% 6.43 合计 / / / / 100.00 氧化后液 2.10 915 / 2.25 / 表 3 不同交换树脂静态吸附情况
Table 3. Static adsorption of different exchange resin
树脂类型 交换前液(1) 2 h交换尾液(1) 2 h吸附容量(2) 吸附率/% 24 h交换尾液(1) 24 h吸附容量(2) 吸附率/% 90 h交换尾液(1) 90 h吸附容量(2) 吸附率/% D202 2.33 0.99 134.00 57.51 0.75 154.80 67.81 0.61 165.07 73.82 D201 2.14 0.48 166.00 77.57 0.28 183.33 86.92 0.12 195.06 94.39 D314 2.14 0.60 154.00 71.96 0.39 172.20 81.78 0.15 189.80 92.99 备注:(1)单位为g/L-V2O5;(2)单位为mg/mL湿树脂。 表 4 静态解吸试验结果
Table 4. Static analytical experimental results
树脂类型 解吸剂 树脂V2O5总含量/mg 解吸液V2O5含量/(g·L-1) 解吸率/% 4%NaOH+4%NaCl 273.54 2.24 99.98 D201 4%NaOH 246.46 1.47 89.24 4%NaCl 238.55 0.11 6.76 -
[1] 杨康. 石煤湿法提钒新工艺与机理研究[D]. 湘潭: 湘潭大学, 2010.
YANG K. A novel method for the vanadium extraction from stone coal[D]. Xiangtan: Xiangtan University, 2010.
[2] 程倩, 王明, 宁新霞, 等. 从某低品位炭质钒矿石中酸浸-萃取-氨沉淀提钒试验研究[J]. 矿产综合利用, 2021(3): 17-21. https://www.cnki.com.cn/Article/CJFDTOTAL-KCZL202103003.htm
CHENG Q, WANG M, NING X X, et al. Process of vanadium extraction from a low-grade carbonaceous vanadium by acid leaching-extraction-ammonia precipitation[J]. Multipurpose Utilization of Mineral Resources, 2021(3): 17-21. https://www.cnki.com.cn/Article/CJFDTOTAL-KCZL202103003.htm
[3] 王明, 程倩, 齐建云, 等. 石煤钒矿硫酸低温熟化-柱浸提钒工艺[J]. 矿冶, 2020, 29(3): 62-67. https://www.cnki.com.cn/Article/CJFDTOTAL-KYZZ202003013.htm
WANG M, CHENG Q, QI J Y, et al. Process of vanadium extraction from tone coal vanadium ore by sulfuric acid low temperature curing and column leaching[J]. Mining and Metallurgy, 2020, 29(3): 62-67. https://www.cnki.com.cn/Article/CJFDTOTAL-KYZZ202003013.htm
[4] 曾添文, 戴文灿, 张志, 等. 离子交换树脂对钒(Ⅴ)交换性能的研究[J]. 离子交换与吸附, 2002, 18(5): 453-458. doi: 10.3321/j.issn:1001-5493.2002.05.011
ZENG T W, DAI W C, ZHANG Z, et al. Study on exchanging properties of anion exchange resins for vanadium (V)[J]. Ion Exchange and Adsorption, 2002, 18(5): 453-458. doi: 10.3321/j.issn:1001-5493.2002.05.011
[5] 蔡晋强. 石煤提钒生产新工艺[J]. 无机盐工业, 2001, 33(5): 37-42. doi: 10.3969/j.issn.1006-4990.2001.05.015
CAI J Q. New technology of extracting vanadium from stone coal[J]. Inorganic Chemical Industry, 2001, 33(5): 37-42. doi: 10.3969/j.issn.1006-4990.2001.05.015
[6] 车荣睿. 离子交换法在治理含钒废水中的应用[J]. 离子交换与吸附, 1994(3): 333-337.
CHE R R. The treatment of waste water containing vanadium ions by ion exchange, 1994(3): 333-337.
[7] 戴文灿, 朱柒金, 陈庆邦, 等. 石煤提钒综合利用新工艺的研究[J]. 有色金属(选矿部分), 2000(3): 15-18. doi: 10.3969/j.issn.1671-9492.2000.03.004
DAI W C, ZHU Q J, CHEN Q B, et al. Study on new technology of comprehensive utilization of vanadium from stone coal[J]. Nonferrous Metals (Mineral processing part), 2000(3): 15-18. doi: 10.3969/j.issn.1671-9492.2000.03.004
[8] 曾理, 李青刚, 肖连生. 离子交换法从石煤含钒浸出液中提钒的研究[J]. 稀有金属, 2007, 31(3): 362-366. doi: 10.3969/j.issn.0258-7076.2007.03.017
ZENG L, LI Q G, XIAO L S. Study of extraction of vanadium from the vanadiferous leaching liquor of rock-coal by ion exchange[J]. Rare Metal, 2007, 31(3): 362-366. doi: 10.3969/j.issn.0258-7076.2007.03.017
[9] 王斌. 石煤浸出液离子交换法提钒的研究[J]. 钢铁钒钛, 2007, 28(1): 22-25. doi: 10.3969/j.issn.1004-7638.2007.01.005
WANG B. Study on extraction of vanadium from acid leaching solution of stone coal with Ion exchange resin[J]. Iron Steel Vanadium Titanium, 2007, 28(1): 22-25. doi: 10.3969/j.issn.1004-7638.2007.01.005
[10] 康兴东, 张一敏, 黄晶, 等. 石煤提钒离子交换工艺研究[J]. 矿产保护与利用, 2008, 39(3): 53-55. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=cc841bb1-4cea-40ec-9031-94739466df7f
KANG X D, ZHANG Y M, HUANG J, et al. Study on extraction process of vanadium from stone coal by ion-exchange[J]. Conservation and Utilization of Mineral Resources, 2008, 39(3): 53-55. http://kcbh.cbpt.cnki.net/WKD/WebPublication/paperDigest.aspx?paperID=cc841bb1-4cea-40ec-9031-94739466df7f
[11] 刘彦华, 杨超. 用D301树脂从含钒萃余液中回收钒的试验研究[J]. 湿法冶金, 2010, 29(2): 96-98. doi: 10.3969/j.issn.1009-2617.2010.02.008
LIU Y H, YANG C. Research on recovery of vanadium from vanadium raffinate using D301 ion-exchange resin[J]. Hydrometallurgy of China, 2010, 29(2): 96-98. doi: 10.3969/j.issn.1009-2617.2010.02.008
[12] 赵坤. 离子交换树脂对钒(Ⅴ)离子的吸附行为及其应用[D]. 长沙: 中南大学, 2010.
ZHAO K. Adsorption Behavior of vanadium (V) ion by ion exchange resin and its application[D]. Changsha: Central South University, 2010.
[13] 冯其明, 孙健程, 张国范, 等. D201树脂吸附钒(V)的过程[J]. 有色金属, 2010(1): 73-76. doi: 10.3969/j.issn.2095-1744.2010.01.018
FENG Q M, SUN J C, ZHANG G F, et al. Adsorption process of vanadium (V) with D201 resin[J]. Nonferrous Metals, 2010(1): 73-76. doi: 10.3969/j.issn.2095-1744.2010.01.018
[14] 段冉. 高纯五氧化二钒的制备及偏钒酸铵结晶机理的研究[D]. 长沙: 中南大学, 2011.
DUAN R. Preparation of high purity V2O5 and study on crystalline of ammonium vanadate[D]. Changsha: Central South University, 2011.
[15] 陈薇. 陕西某石煤提钒试验与机理研究[D]. 昆明: 昆明理工大学, 2008.
CHEN W. Experiment and mechanism study on vanadium extraction from a stone coal in Shaanxi province[D]. Kunming: Kunming University of Science and Technology, 2008.
-



 下载:
下载: