Abnormal Hot Blank of Oxygen-free Copper Sample Holder and Implications for Laser 40Ar/39 Ar Dating
-
摘要:
激光40Ar/39Ar定年方法中扣除的本底是样品测试过程中的系统冷本底。在满足激光加热样品时样品盘升温幅度有限和样品盘已完全脱气两个条件的情况下,这一处理方式的有效性才能得到保证。本文利用具有不同大气暴露史的无氧铜样品盘结合透长石标准样品YBCs,使用相同的脱气及测试流程,对比分析了不同样品盘的冷、热本底以及放置于不同样品盘时YBCs的大气氩含量。分析结果表明,放置于暴露大气14个月的样品盘内时,YBCs透长石大气氩含量高达约34.4%,使用预先激光去气的样品盘此值可降低至约2%;暴露大气约10个月的样品盘,激光加热其两个样品孔时,40Ar脱气量可达约1.6×10−14~3.1×10−14mol;暴露时长约为26个月的样品盘,40Ar含量升高至约0.8×10−13~2.0×10−13mol;它们均远高于系统冷本底3.8×10−16~6.2×10−16mol。两个样品盘热本底40Ar/36Ar值约为310,高于大气氩比值。因此,对于暴露大气时间较长的样品盘,约150℃去气四天的流程不足以使其完全脱气。激光加热样品时会导致样品盘局部升温,脱气不完全的样品盘会释放出大量热本底。模拟以及标准样品测试均显示了这种情况会影响辐照参数J值以及年龄的计算。激光微量年轻样品40Ar/39Ar定年过程中,建议装样后对无氧铜样品盘进行300~400℃至少5h的预脱气,以保证测试数据质量。实验室不具备预脱气条件时,持续使用同一样品盘也可以有效地降低异常热本底对测试结果的影响。
-
关键词:
- 激光40Ar/39Ar定年 /
- 无氧铜样品盘 /
- 热本底 /
- 脱气 /
- 质谱分析
Abstract:BACKGROUND Oxygen-free copper (OFC) tray is usually used as a container for samples of laser 40Ar/39Ar dating. The blank of the mass spectrometry system needs to be subtracted from the sample signal before age calculation. The tray’s hot blank of argon, including its amount and isotope ratio will affect the 40Ar/39Ar age calculation. However, the material and structure of the laser window make the laser chamber susceptible to temperatures higher than 150°C during the degassing procedure of laser 40Ar/39Ar dating[4,10]. This temperature may be too low to degas the OFC tray completely during the experiment. Besides, the wells loading the mineral samples limit the direct acquisition of the hot blank of the system. These two points make it very difficult to accurately deduct the blank signal of laser 40Ar/39Ar dating.
OBJECTIVES To testify to the effectiveness of the traditional degassing procedure on the sample holder and evaluate the effect of hot blank on age calculation of laser 40Ar/39Ar dating.
METHODS To confirm the effectiveness of the degassing procedure, the hot blank of OFC trays, which had been exposed to atmosphere for different time intervals were measured by mass spectrometry, after degassing the laser chamber to 150℃ for four days. Loading the standard mineral YBCs sanidine in the well in OFC trays with different duration in air, the argon isotopes were measured after the same degassing procedure. The change of the proportion of atmospheric argon of YBCs will verify if laser energy heats the OFC tray and mineral sample simultaneously.
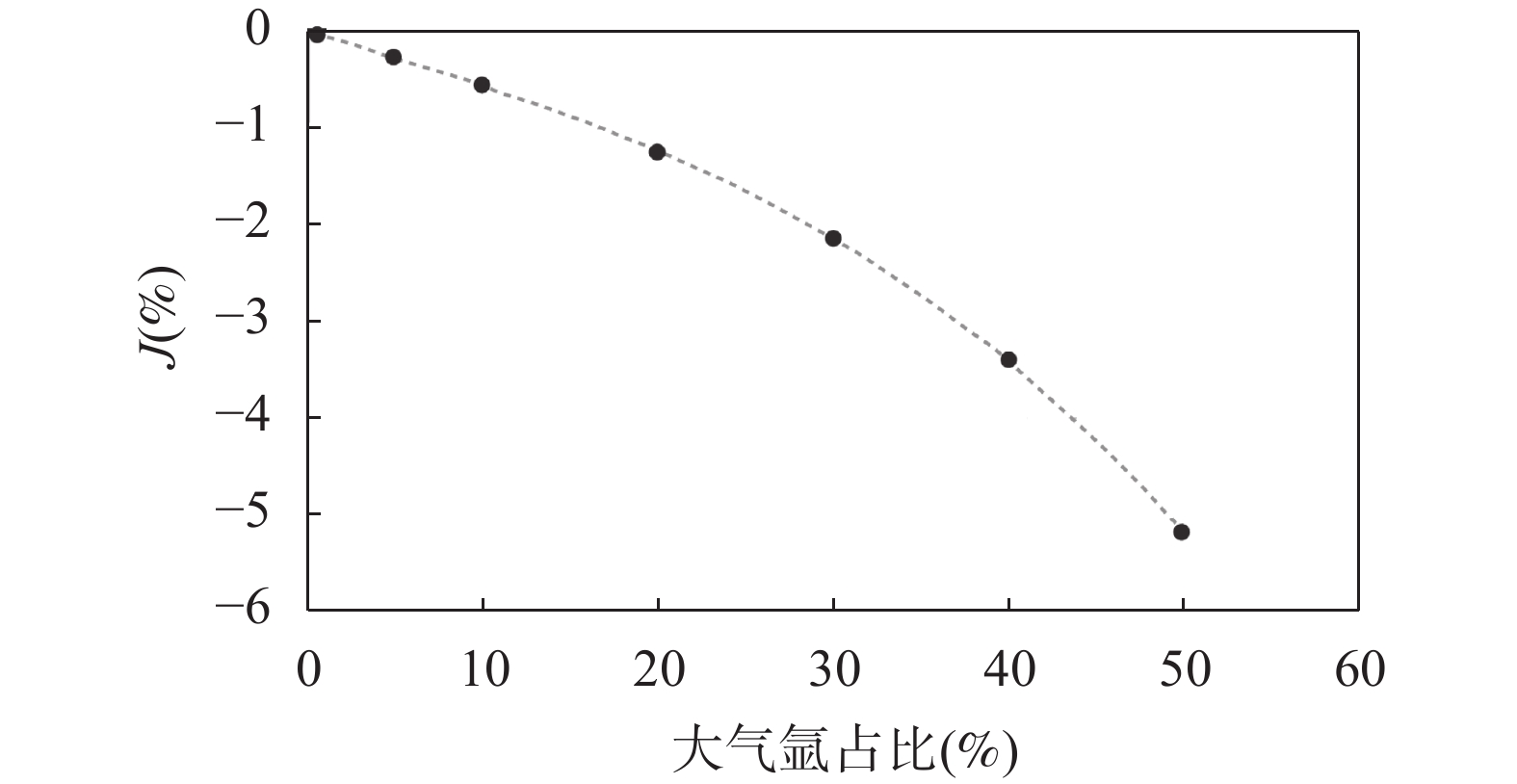
RESULTS The temperature of the sample holder increases during laser heating of samples. For standard mineral YBCs sanidine placing in OFC tray C, exposed to the atmosphere for about 14 months, the proportion of atmospheric argon was 34.4% (Table 1). This value decreased to 2.0% when YBCs was placed in tray D and degassed by laser at higher energy. This implies that the tray temperature will increase during sample heating by laser, and a considerable amount of gas will be released if the tray is not completely degassed. It was proved that heated to about 150℃ was not enough to completely degas the OFC tray during experiment. The hot blank of another two planchettes following the same degassing procedure were measured. For tray A with exposure to the atmosphere for 10 months, the amount of 40Ar released from two wells reached 1.6×10−14−3.1×10−14mol; for tray B with exposure to the atmosphere for 26 months, the 40Ar content increased to 0.8×10−13−2.0×10−13mol. These levels were much higher than the cold background of 3.8×10−16−6.2×10−16mol. Incomplete degassing of the tray may lead to argon isotopic fractionation, resulting in the value of 40Ar/36Ar of the hot blank rise to 310, which is higher than the value of atmospheric argon. Under this condition, using 40Ar/36Ar=295.5[16] or 298.56[17] to correct the atmospheric argon will add extra 40Ar to the sample’s signal and lead to an older age. The OFC tray was degassed efficiently while heated with a higher laser power. Under the same degassing procedure, the hot blank of the empty wells in tray D was similar to the cold blank of the system. The OFC trays exposed to air also gave the same conclusion. When the wells in the tray were heated by a lower power energy following a higher power energy, the hot blank dropped to the cold blank level (Fig.2). Assuming all of the atmospheric argon contributed from the hot blank with 40Ar/36Ar=310, its effect on 40Ar/39Ar age calculation with different combination of 40Ar*/39ArK, J-value and atmospheric argon content was calculated. Results show that the percent change of age(t%) was mainly controlled by the atmospheric argon content. When the atmospheric argon content increased to 50% (Fig.3), it elevated the age by 5%. Under the same condition, the J-value was about 5.2% lower(Fig.4). In practice, however, the different capacity of adsorption and thermal desorption of gas of different minerals[18-19] makes the effects difficult to quantify.
CONCLUSIONS Careful degassing of the OFC tray is needed, especially in very young samples dating through the laser heating method. The temperature of the sample holder increases when the sample is heated by laser, releasing considerable amounts of gas if the holder is incompletely degassed. The limited temperature of the degassing procedure cannot degas the sample holder completely, if the holder adsorbs a lot of gases. There are two possible solutions to solve this problem. The first is pre-degassing the sample holder at 300-400℃ for at least 5 hours at high vacuum furnace after sample exchange. The second is reusing the same sample holder continuously in laser 40Ar/39Ar dating. A combination of both solutions is likely to be most effective.
-

-
表 1 透长石YBCs样品A5及A5-1测试结果对比:A5显示样品各阶步均含有大量大气氩,A5-1大气氩含量正常(初始氩校正采用40Ar/36Ar=295.5)
Table 1. Result comparison of sanidine YBCs. A5 placed on an OFC tray exposures to air for fourteen months; A5-1 on a reusing tray; data calculation under 40Ar/36Ar=295.5. The proportion of atmospheric argon of steps of A5 is high, and return to normal of A5-1.
透长石YBCs样品A5 激光能量
(W)是否可以形成
坪的阶步40Ar/39Ar 36Ar/39Ar 40Ar*/39Ark 40Ar*(%) 辐照剂量监测 J值 ±2σ 0.24 - 10.319367 0.016838 5.34346 51.78 0.0030628 ±0.0000796 0.39 - 7.088052 0.006638 5.12619 72.32 0.0031926 ±0.0000388 0.54 - 8.306639 0.010221 5.28589 63.63 0.0030961 ±0.0000489 0.70 - 7.192548 0.007138 5.08307 70.67 0.0032197 ±0.0000417 1.32 - 7.205940 0.007393 5.02091 69.68 0.0032595 ±0.0000454 全熔 - - - - 65.59 0.0031648 ±0.0000226 透长石YBCs样品A5-1 激光能量
(W)是否可以形成
坪的阶步40Ar/39Ar 36Ar/39Ar 40Ar*/39Ark 40Ar*(%) 辐照剂量监测 J值 ±2σ 0.16 - 5.363579 0.000023 5.35657 99.87 0.0030553 ±0.0000345 0.39 √ 5.127897 0.000060 5.10986 99.65 0.0032028 ±0.0000205 0.54 √ 5.194142 0.000406 5.07372 97.68 0.0032256 ±0.0000205 0.70 √ 5.312808 0.000752 5.09012 95.81 0.0032152 ±0.0000204 1.32 √ 5.186170 0.000242 5.11425 98.61 0.0032000 ±0.0000194 全熔 - - - - 97.95 0.0032045 ±0.0000106 注:“√”表示可以形成坪的阶步;“-”表示不可以形成坪的阶步。 -
[1] McDougall I, Harrison T M. Geochronology and thermochronology by the 40Ar/39Ar method (The second edition)[M]. New York: Oxford Uninversity Press, 1999: 81-82.
[2] 张佳, 刘汉彬, 李军杰, 等. K-Ar稀释法中40Ar含量测量过程中实验参数的确定[J]. 岩矿测试, 2021, 40(3): 451−459.
Zhang J, Liu H B, Li J J, et al. Determination of experimental parameters during measurement of 40Ar content in K-Ar dilution method[J]. Rock and Mineral Analysis, 2021, 40(3): 451−459.
[3] Shi W B, Wang F, Wu L, et al. Geologically meaningful 40Ar/39Ar ages of altered biotite from a polyphase deformed shear zone obtained by in vacuo step-heating method: A case study of the Waziyü detachment fault, Northeast China[J]. Minerals, 2020, 10: 648.
[4] Wang F, He H Y, Zhu R X, et al. Laser step-heating 40Ar/39Ar dating on young volcanic rocks[J]. Chinese Science Bulletin, 2006, 51(23): 2892−2896. doi: 10.1007/s11434-006-2195-9
[5] Barfod D N, Mark D F, Tait A, et al. Argon extraction from geological samples by CO2 scanning laser step-heating[J]. London Geological Society (Special Publications), 2014, 378: 79−90. doi: 10.1144/SP378.23
[6] 高梓涵, 李立武, 王玉慧, 等. 双真空炉管的研制及其在岩石加热脱气气体组分测试中的应用[J]. 岩矿测试, 2019, 38(5): 469−478.
Gao Z H, Li L W, Wang Y H, et al. Development of a double vacuum furnace tube and its application in gas composition determination during rock heating degassing[J]. Rock and Mineral Analysis, 2019, 38(5): 469−478.
[7] 张万峰, 邱华宁, 郑德文, 等. 40Ar/39Ar定年自动去气系统的研制及其性能[J]. 地球化学, 2020, 49(5): 509−515.
Zhang W F, Qiu H N, Zheng D W, et al. An automatic degassing system for 40Ar/39Ar dating[J]. Geochimica, 2020, 49(5): 509−515.
[8] Wang F, Shi W B, Zhang W B, et al. Multiple phases of mountain building on the Northern Xizang margin[J]. Lithosphere, 2020: 8829964.
[9] 邱华宁. 新一代Ar-Ar实验室建设与发展趋势: 以中国科学院广州地球化学研究所Ar-Ar实验室为例[J]. 地球化学, 2006, 35(2): 133−140. doi: 10.3321/j.issn:0379-1726.2006.02.003
Qiu H N. Construction and development of new Ar-Ar laboratories in China: Insight from GV-5400 Ar-Ar laboratory in Guangzhou Institute of Geochemistry, Chinese Academy of Sciences[J]. Geochimica, 2006, 35(2): 133−140. doi: 10.3321/j.issn:0379-1726.2006.02.003
[10] McIntosh W C, Heizler M T. Applications of CO2 laser heating in 40Ar/39Ar geochronology[C]//Lanphere M A, Dalrymple G B, Turrin B D. Eighth International Conference on Geochronology, Cosmochronology and Isotope Geology. USA: US Geological Survey Circular 1107, 1994: 212.
[11] Wang F, Jourdan F, Lo C H, et al. YBCs sanidine: A new standard for 40Ar/39Ar dating[J]. Chemical Geology, 2014, 388: 87−97. doi: 10.1016/j.chemgeo.2014.09.003
[12] Koppers A A P. ArArCALC-software for 40Ar/39Ar age calculations[J]. Computers & Geosciences, 2002, 28: 605−619.
[13] Steiger R H, Jäger E. Subcommision on geochronology: Convention on the use of decay constants in geo- and cosmochronology[J]. Earth and Planetary Science Letters, 1977, 36: 359−362. doi: 10.1016/0012-821X(77)90060-7
[14] 杨列坤, 王非, 贺怀宇, 等. 年轻火山岩氩同位素体系定年技术最新进展及问题[J]. 地震地质, 2009, 31(1): 174−185. doi: 10.3969/j.issn.0253-4967.2009.01.016
Yang L K, Wang F, He H Y, et al. Achievements and limitations of 40Ar/39Ar dating on young volcanic rocks[J]. Seismology and Geology, 2009, 31(1): 174−185. doi: 10.3969/j.issn.0253-4967.2009.01.016
[15] 高本辉, 李林. 金属片高温出气[J]. 电子管技术, 1978(2): 136−143.
Gao B H, Li L. The sheet metal outgassing characteristics at high temperature[J]. Evacuated Tube Technology, 1978(2): 136−143.
[16] Renne P R, Cassata W S, Morgan L E. The isotopic composition of atmospheric argon and 40Ar/39Ar geochronology: Time for a change?[J]. Quaternary Geochronology, 2009, 4: 288−298. doi: 10.1016/j.quageo.2009.02.015
[17] Nier A O. A redetermination of the relative abundances of the isotopes of carbon, nitrogen, oxygen, argon, and potassium[J]. Physical Review, 1950, 77: 789−793. doi: 10.1103/PhysRev.77.789
[18] Lee J Y, Marti K, Severinghas J P, et al. A redetermination of the isotopic abundances of atmospheric Ar[J]. Geochimica et Cosmochimica Acta, 2006, 70: 4507−4512. doi: 10.1016/j.gca.2006.06.1563
[19] Wang F, Shi W, Guillou H, et al. A new unspiked K−Ar dating approach using laser fusion on microsamples[J]. Rapid Communications in Mass Spectrometry, 2019, 33: 587−599. doi: 10.1002/rcm.8385
[20] Phillips D, Matchan E L, Honda M, et al. Astronomical calibration of 40Ar/39Ar reference minerals using high-precision, multi-collector (ARGUSVI) mass spectrometry[J]. Geochimica et Cosmochimica Acta, 2017, 196: 351−369. doi: 10.1016/j.gca.2016.09.027
[21] Velthaus V, Tietz B, Trautmann C, et al. Desorption measurements of accelerator-related materials exposed to different stimuli[J]. Vacuum, 2021, 194: 110608. doi: 10.1016/j.vacuum.2021.110608
-




 下载:
下载: